Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
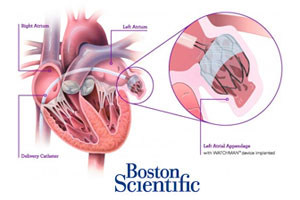
3. FDA wants another panel hearing on Boston Scientific’s Watchman anti-stroke device
Boston Scientific CFO Dan Brennan said today that the FDA wants to hold another advisory panel hearing on its Watchman anti-stroke device and pushed the approval timeline for the Watchman heart implant into next year. Brennan, speaking today at the Wells Fargo Healthcare Conference, said Boston Scientific is now hoping for an approval during the 1st half of 2015.
The as-yet-unscheduled panel would be the 3rd advisory committee hearing over Boston Scientific’s pre-market approval application for the Watchman device and comes despite positive votes from the 2 prior meetings. Read more
![]()
![]()
2. Medtronic’s OUS move becomes a political football

MASSDEVICE ON CALL – Medtronic’s acquisition of Covidien has spurred hundreds (if not thousands) of articles, analyses and speculations but at least one political hopeful is using the blockbuster news to gain favor in Medtronic’s home state.
Minnesota Republican and candidate for governor Marty Seifert issued a press release on Sunday blaming the Obama Administration for chasing Medtronic out of the U.S. with the medical device tax. Seifert is running for office against incumbent Mark Dayton (D), who has held the governor’s office since 2011. Read more
![]()
1. Medtronic buyout holds $80M in golden parachutes for Covidien execs
If the top executives at Covidien decide to leave the company after a $43 billion acquisition by Medtronic goes through, they’ll do it under a golden parachute worth a collective $80 million, regulatory filings show.
The top 5 “named executives” at Covidien, led by chairman, president & CEO Joe Almeida, are in line for an average payout of some $16 million if they elect to depart after the deal’s consummation, slated for the end of this year or early next year. Read more