Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
3. Boston Scientific can’t sever Pinnacle pelvic mesh cases
Boston Scientific last week lost a bid to remove 4 product liability lawsuits from the multi-district litigation filed over its Pinnacle pelvic mesh device when a federal judge dismissed its motion to sever and applied the ruling to all past and future cases added to the MDL.
The Marlborough, Mass.-based medical device company as of August 5 was facing more than 23,000 similar lawsuits in state and federal courts, Boston Scientific said in a regulatory filing this month. Some 1,700 have been consolidated under a Massachusetts state court, the company said; the federal cases have been gathered into an MDL under Judge Joseph Goodwin of the U.S. District Court for Western Virginia. Read more
![]()
![]()
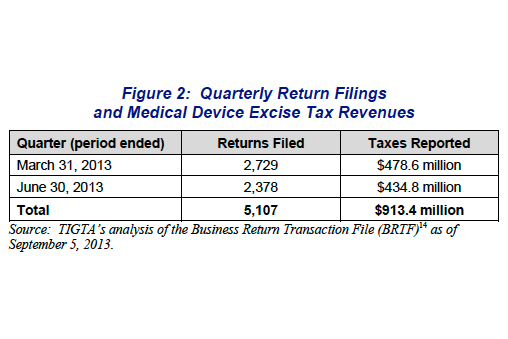
2. Medtech tax slip-up fuels calls for repeal
Medical device industry lobbyists have new ammunition in the fight against the 2.3% Affordable Care Act tax thanks to a U.S. Treasury report citing several issues with the IRS’s management of the levy.
Tax regulators are apparently having trouble determining who does and does not owe the tax and the confusion has also spurred hundreds of false penalties issued to companies wrongly asked to pay, according to the report. Read more
![]()
1. Covidien snaps up Reverse Medical
Covidien isn’t letting its impending acquisition by Medtronic slow down its own merger activity, announcing that it acquired Reverse Medical and its line of vascular devices for an unspecified amount.
The move adds Reverse Medical’s CE-Marked Barrel brain aneurysm device, UNO neurovascular embolization system and MVP micro-vascular plugs to Mansfield, Mass.-based Covidien’s neurovascular segment. Read more