Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
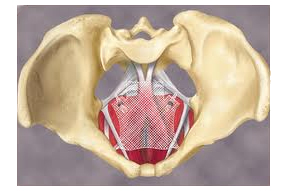
3. Plaintiffs look to remove pelvic mesh cases to Pennsylvania state court
A group of plaintiffs who are suing Boston Scientific, Johnson & Johnson, Secant Medical and other pelvic mesh makers want a federal judge to send their cases back to a state court in Philadelphia.
Boston Scientific, J&J’s Ethicon subsidiary, C.R. Bard, Endo Health Solutions, Cook Medical and Coloplast are facing thousands of product liability and personal injury lawsuits over their respective pelvic mesh devices. Hundreds of cases have been consolidated into multi-district litigation under Judge Joseph Goodwin of the U.S. District Court for Southern West Virginia. Read more
![]()
![]()
2. Is there an Obamacare silver lining for medtech?

MASSDEVICE ON CALL — Wall Street analyst and Motley Fool writer Sean Williams sees an Obamacare upside for the medical device industry, despite the doom and gloom forecast usually spurred by talk of the medical device tax.
Williams ceded that his opinion is “pure conjecture,” but he speculated that the late surge in Obamacare enrollment may suggest a financial benefit to healthcare systems and thus an indirect boon to medical device companies as hospitals write off fewer bills as “noncollectable” and spend more on new technologies. Read more
![]()
1. GE Healthcare lands FDA nod for all-in-one CT
GE Healthcare’s this week touted a new FDA win for its latest in CT scanner technology, saying the Revolution* CT^ is the 1st of its kind that’s right for “every clinical specialty.”
The healthcare giant landed 510(k) clearance to market the device as an all-in-one CT scanner for cardiology, oncology and neurology, saying the sedation-free imaging technology could play an important role for “sensitive” patient groups, such as pediatric, renal insufficiency, trauma and stroke patients. Read more