
Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
3. Medicare proposes TAVI reimbursement guidelines
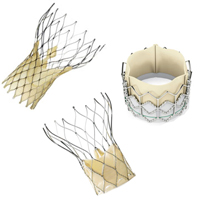
The Centers for Medicare & Medicaid Services released a proposal detailing guidelines for reimbursement of transcatheter aortic valve implantation procedures for treating severe symptomatic aortic stenosis. The memo defines 5 requirements for coverage, including the types of facilities that may apply for reimbursement and enrollment in post-procedure registries to track patient outcomes.
![]()
![]()
2. Boston Scientific to hit the acquisitions trail
Boston Scientific (NYSE:BSX) plans to hoard most of the more than $1 billion in free cash flow it expects to generate this year so it can make acquisitions to boost its top line, CFO Jeff Capello told analysts. The Natick, Mass.-based medical device giant reported 4th-quarter profits of $107 million, or 7 cents per diluted share, on sales of $1.85 billion for the 3 months ended Dec. 31, 2011.
![]()
1. IRS releases guidance on 2.3% levy, Paulsen calls for a vote on repeal

The U.S. Department of the Treasury released proposed regulations on who will and who won’t be subject to the 2.3% medical device tax, carving out some exemptions for early-stage devices and providing a little clarity on fears of a double tax for contract manufacturers, importers and component manufacturers.

