
Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
3. Philips touts HeartNavigator clearance
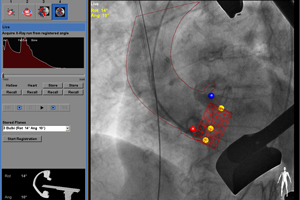
Philips Healthcare (NYSE:PHG) landed FDA clearance for its HeartNavigator heart valve replacement system. Medical City Dallas Hospital became the first U.S. hospital to use the system, which was launched in Europe in the first quarter of this year.
![]()
Sign up to get our free newsletters delivered straight to your inbox
![]()
2. Congress agrees to 2-month “doc fix”

House Speaker John Boehner (R-Ohio) said that the House will pass a 2-month extension to the "doc fix," a payroll tax break extension saving doctors from a looming cut in Medicare reimbursement payments.
![]()
1. Former CDRH director joins Public Citizen in demanding recall of Stryker’s brain stent
Former CDRH director Larry Kessler joined consumer watchdog group Public Citizen asking the federal government to recall Stryker Corp.’s (NYSE:SYK) Wingspan brain stent over concerns that it may lead to heightened risk of stroke and death.

