
Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
3. Med-tech for paralysis patients wins key radio spectrum

Medical devices that help patients regain functionality and sensation in paralyzed limbs landed a win with government approval of a key radio frequency tuned to transmitting signals through the body.
![]()
Sign up to get our free newsletters delivered straight to your inbox
![]()
2. Study reinforces stent crumpling concern
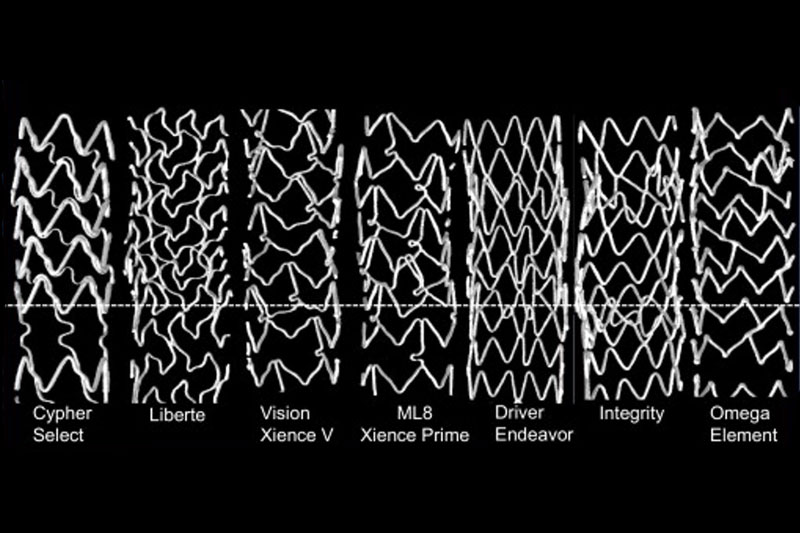
The thinner design of new stents such as Boston Scientific’s (NYSE:BSX) Element and Omega models and Medtronic’s (NYSE:MDT) Driver warp more easily warped than competing devices, according to bench-test results from seven stent models.
![]()
1. Diabetes: FDA releases artificial pancreas guidance

The FDA released a long-awaited draft guidance on an approval pathway for "artificial pancreas" devices, a breakthrough treatment for patients with Type I diabetes.

