Medical device industry giant Medtronic (NYSE:MDT) is back in court this month for a re-hearing of a patient injury trial accusing the company of wrongdoing that left an Arizona man paralyzed.
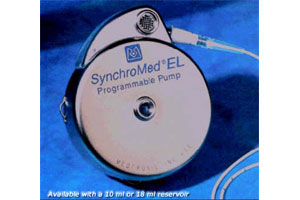
Lawyers traded barbs in court this week as the plaintiff argued that Medtronic had failed to properly provide product updates the FDA that would have earlier warned physicians and patients about potential issues with the medical device company’s SynchroMed programmable pain pump implant.
The case had been thrown out of court earlier this year when a federal judge dismissed the case on grounds of grounds of preemption, which holds that patients can’t sue medical device makers in state courts over products that went through the FDA’s most stringent review process, called pre-market approval.
In July the U.S. Court of Appeals for the 9th Circuit granted en banc rehearing of the case, which was conducted this week. Lawyers for the plaintiff argued that Medtronic failed to uphold its responsibilities to warn the public about potential medical device defects.
"If what should have occurred had occurred, an earlier warning would have gone out," patient lawyer Tom Cotter said in court.
Medtronic’s lawyers insisted that each of the plaintiff’s complaints were preempted, the Courthouse New Service reported.
"At the end of the day what they’re asking is that there be a warning that is different from or addition to the federally mandated warning," defending attorney Michael Brown argued.
Chief Judge Alex Kozinski disagreed with Medtronic’s attorney.
"I didn’t hear that," Kozinski said. "Their claim is that your client should have notified the FDA sooner of these problems. And, had they done that, word would have gotten out, either by osmosis or by the FDA taking steps to advise them to warn. That’s the claim, not that they had to anything more than the FDA required but that they had to do it sooner."
In 2000, plaintiff Richard Stengel began using Medtronic’s SynchroMed EL pain pump, an implanted device that delivers medication directly to the spine via an intrathecal catheter. In 2005, Stengel began developing symptoms of paralysis from an inflammation in his spine around the catheter tip. The device and most of the inflammation were removed, "but not in time to prevent the granuloma from rendering Stengel permanently paraplegic," according to court documents.
The FDA approved the first SynchroMed pump in 1998 and later granted approval for the iteration used to treat Stengel. Medtronic recalled that device in 2008.

