No can do: First-in-human study evaluates ‘implantable string’ S-ICD
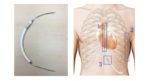 An “implantable string” S-ICD developed by Israel’s Newpace Medical was implantable in about 20 minutes and could offer lower infection rates and cosmetic benefits, according to a small, 1st-in-human study presented at HRS 2017.
An “implantable string” S-ICD developed by Israel’s Newpace Medical was implantable in about 20 minutes and could offer lower infection rates and cosmetic benefits, according to a small, 1st-in-human study presented at HRS 2017.
The ISSD removes the necessity for a “can” to hold the impluse generator in conventional and S-ICD devices, “where the ICD components and functionality are compound into a single, flexible, string-shape device,” according to the study, which sought to show that the coil would have enough surface area to deivler effective therapy.
The ISSD was implanted in 22 patients; preliminary results showed an average defibrillation testing threshold of 26J and an SD of 11J.
“This defibrillation method with a flexible, can-less defibrillator that can be placed without the need for a surgical pocket may allow improved patient compliance and aesthetics, be less invasive, and shorter implantation time. In May 2017, full analysis data will be available on the study,” the authors wrote.
“The results are very promising, because not only does the subcutaneous string device provide a minimally invasive approach that lowers the risk of infection for patients, but we were also able to successfully implant the device in a short amount of time when compared to other defibrillators,” lead author Dr. Petr Neužil, of Prague’s Na Homolce Hospital, said in prepared remarks. “We’re optimistic about this innovative technology and what it means for the future of healthcare. It’s an example of how advancing technology has the potential to improve the quality of life for patients and, ultimately, improve patient outcomes.”

