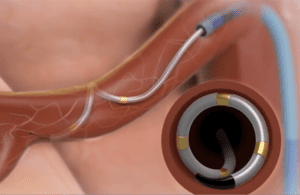
The panel unanimously said the minimally invasive catheter system is safe, but was nearly split on efficacy. With six members voting that the benefits outweighed the risk, six voting the other way and one panelist abstaining, panel chair Dr. Richard Lange cast the deciding vote to recommend against FDA approval.
It’s not necessarily the end for Medtronic’s effort to secure approval of its Symplicity Spyral multi-electrode RDN catheter and Symplicity G3 radiofrequency generator to reduce blood pressure in certain hypertension patients. From here, the FDA will consider the panel`s recommendation and let Medtronic know whether it agrees and whether additional information is needed.
“We’re looking forward to working together with the FDA and taking into account all of the comments that we heard today,” Medtronic SVP and Chief Scientific, Medical, and Regulatory Officer Dr. Laura Mauri said after the vote.
The stakes are high for Medtronic, which has been developing the minimally invasive technology for years in the belief that it could be a billion-dollar business and provide relief to millions of patients worldwide, reducing heart attacks, strokes and other serious events tied to high blood pressure.
The same FDA review panel yesterday supported approval of competing RDN technology developed by Otsuka Medical Devices’ ReCor Medical. If the FDA agrees with the panel’s recommendation on the safety and efficacy of ReCor’s system, it could be the first RDN therapy to be approved for use in the U.S.
ReCor’s clinical trial met its primary endpoint for efficacy, while Medtronic’s RDN trials have failed to hit that target — though analysts have said they don’t think that will prevent Medtronic from winning FDA approval.

