Gaining the latest EUAs were machines by Medtronic (NYSE:MDT), Draeger, MEK-ICS, Ventec Life Systems, and startup CoLabs.
One of those EUAs went to Medtronic’s Puritan Bennett 560 ventilator, the same machine whose design the company made public in March. Fridley, Minn.-based Medtronic said yesterday that it expects the PB560 will be available in the U.S. in May. Introduced in 2010 and currently sold in 35 countries around the world for an average price of $10,000 apiece. Medtronic described the PB560 as a compact, lightweight, and portable ventilator that can be used in clinical settings and at home.
Medtronic also said the open-source plans for the PB560 have been downloaded 90,000 times, and that it has made “meaningful progress” with three open-source manufacturers, including Baylis Medical in Ontario, Canada, Taiwan-based Foxconn Technology Group at its plant in Mount Pleasant, Wis., and Vingroup in Hanoi, Vietnam.
Fridley, Minn.-based Medtronic also said it has deployed remote management capability of its Puritan Bennett 980 ventilator to two unidentified U.S. hospitals, with help from Intel. The new capability will enable clinicians to adjust the ventilator settings outside of the intensive care unit and away from the patient, which may reduce healthcare worker and clinician exposure to patients recovering from the virus. If the feature is well received, the company said it may offer it to other U.S. customers later in April.
In all, Medtronic said it is shipping more than 300 ventilators per week to customers in the the highest-risk, highest-need locations in the world, with a focus on the PB980 and PB560 models. The company expects to ship more than 400 ventilators per week by the end of April, over 700 per week by the end of May and more than 1000 per week by the end of June. Over the next 6 months, Medtronic expects to produce more than 25,000 ventilators.
Other medical equipment that received FDA emergency authorization this week included:

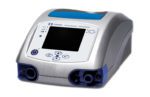
- South Korean medtech manufacturer MEK-ICS‘ MTV 1000 portable ventilator, which features turbine, oxygen blender and optional pulse oximetry and end tidal carbon dioxide testing functions, according to the company’s website.
- The Draeger Atlan 350 and 350 XL anesthesia gas machines, which the FDA said can be used for mechanical or manual ventilation, pressure-supported spontaneous breathing and spontaneous breathing. The A350/A350 XL is equipped with an electronic gas mixer with automatic fresh-gas composition control, according to Lubeck, Germany-based Draeger.
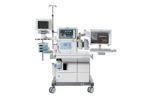
Draeger Atlan 350 ventilator (Image from Draeger) - Ventec Life Systems‘ V+Pro emergency ventilator. General Motors will manufacture Ventec’s FDA-cleared VOCSN critical care ventilators at the automaker’s Kokomo, Ind., plant, with plans to begin shipping this month. Ventec said it was also “taking aggressive steps” to increase production at its plant in Bothell, Wash.
- Startup CoLabs‘ emergency-use Ventor ventilator, which the FDA said consists of a reusable console and a single-use, disposable breathing circuit. Carmel, Calif.-based CoLabs said on its website that Ventor president Clay Nolan describes as “the AED of emergency airway and ventilation.” The machine uses a novel ventilation technique that the company says is the first of its kind to increase blood flow during cardiopulmonary resuscitation.

