Natick, Mass.-based medical device company Corindus Vascular Robotics won FDA clearance for the 1st robot-assisted surgical system designed for percutaneous coronary intervention, the CorPath 200.
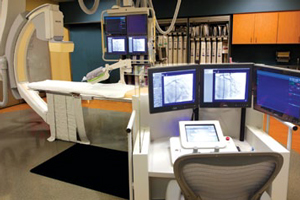
The CorPath 200 is the "1st and only robotic-assisted procedure to allow for controlled placement of coronary guidewires and stent/balloon catheters from an optimized interventional cockpit," according to the company.
In using the system, the surgeon operates CorPath 200 from behind a radiation-shielded "cockpit." Rather than standing over a patient while clad in a lead apron, the operator is seated behind an operating station, manipulating surgical tools with a series of touch-screen and joystick controls.
That reduces a physicians exposure to radiation while also providing a more comfortable and ergonomic experience, especially during long and complex procedures.
"There has been a tremendous amount of interest in the CorPath 200 System, and we are excited to bring to market a solution that has the potential to transform the standard of care for PCI procedures," president & CEO David Handler said in prepared remarks. "This is an important milestone for interventional cardiology as we take the next step in transforming the way these procedures are performed in the future."
In its 1st-in-human studies, which included 8 patients, the robot demonstrated 100% clinical success with no major adverse events at patient discharge after 30 days.
In March 2011 the device maker began clinical trials in support of its 510(k) bid with a 164-patient group, placing coronary stents in procedures performed at 9 sites across the U.S.
CorPath 200 joins a growing arena of robot assisted surgical devices, including Intuitive Surgical’s (NSDQ:ISRG) da Vinci robot and Mazor Robotics’ (PINK:MZRTF) Renaissance device.

