Boston Scientific‘s (NYSE:BSX) Watchman anti-stroke device finally won FDA approval last week, 6 years after an FDA advisory panel 1st recommended approval for the heart implant.
Marlborough, Mass.-based Boston Scientific said March 13 that the pre-market approval nod is for high-risk patients with non-valvular atrial fibrillation who are seeking an alternative to long-term warfarin therapy. Watchman is slated to hit clinics already involved in clinical trials, followed by more sites as physicians are trained up on implanting Watchman.
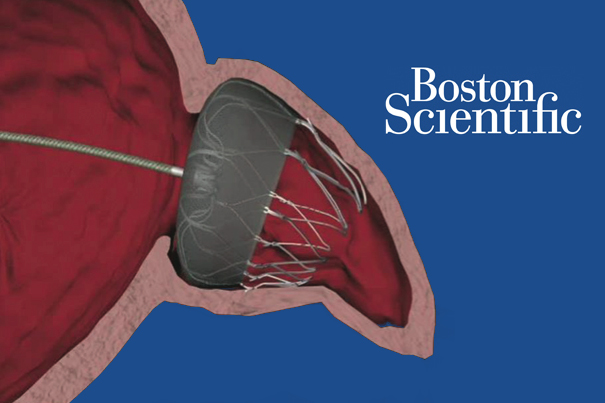
Watchman is a percutaneously delivered cardiac device permanently implanted in the heart’s left atrial appendage to capture clots that may form there and cause a stroke.
Back in 2009 the FDA’s Circulatory Devices panel voted to recommend approval, but the federal safety watchdog issued a "not approvable" letter for Watchman the next year. A 2nd panel voted 13-1 in 2013 that the Watchman implant meets safety and efficacy goals, but last year the FDA said it wanted yet another panel to review the device. The 3rd panel again recommended approval, but by a very narrow margin.
"The Watchman device is an important step forward in stroke management for patients with AF," co-principal investigator Dr. Vivek Reddy of New York City’s Mount Sinai Medical Center said in prepared remarks. "We know that up to 40% of patients who are eligible for oral anticoagulation do not take it for numerous reasons, highlighting the need for additional treatment options. The Watchman device is a breakthrough treatment providing those patients who are suitable for warfarin with an implant-based alternative to long-term warfarin therapy while still reducing the risk of stroke."
"Today marks a defining moment in the company’s journey towards establishing left atrial appendage closure therapy in the United States. Boston Scientific is proud to offer this potentially life-changing stroke risk treatment option to high-risk patients with AF who have a reason to seek a non-drug alternative to warfarin. This therapy could free them from the challenges of long-term warfarin therapy," added Boston Scientific Rhythm Management president Joe Fitzgerald. "FDA approval of the Watchman device is another example of Boston Scientific delivering on its commitment to bring meaningful innovations to patient care."