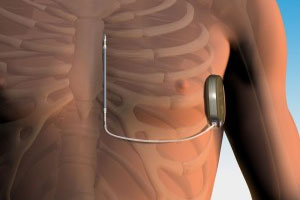
Boston Scientific (NYSE:BSX) and newly acquired Cameron Health won FDA approval for the world’s only commercially available lead-free implantable cardioverter defibrillator, the S-ICD.
Implanted just under the skin, the S-UCD is the only device of its kind that does not require wires threaded into the heart in order to provide therapy. The less-invasive device opens up the technology to a larger swath of patients.
Earlier this year an FDA expert panel voted 7-to-1 in favor of approval for the device, which Boston Scientific acquired in a merger with Cameron Health that’s worth up to $1.35 billion.
"With the addition of the S-ICD System, we believe Boston Scientific has a compelling and highly differentiated portfolio that will help fuel our growth strategy," Boston Scientific interim CEO Hank Kucheman said in prepared remarks. "We are the only company to offer an FDA-approved subcutaneous implantable defibrillator and expect this to be the case for several years."
The S-ICD system won CE Mark approval in the European Union in 2009 and has been on the market outside for some time. More than 1,400 devices have been implanted in patients around the world to date, according to a press release.
Boston Scientific expects to begin a phased launch of the S-ICD system that will expand over the time, the company announced.
"The S-ICD System provides an alternative for treating patients with life-threatening heart arrhythmias for whom the routine ICD placement procedure is not ideal," FDA Center for Devices & Radiological Health’s Office of Device Evaluation director Christy Foreman said in a prepared statement. "Some patients with anatomy that makes it challenging to place one of the implantable defibrillators currently on the market may especially benefit from this device."
That’s more good news for Boston Scientific as it continues gaining milestone in line with incoming CEO Mike Mahoney’s 2012-2013 development and regulatory approval time lines, which he shared during a conference call earlier this year.
"The S-ICD System, coupled with our numerous recent regulatory approvals and our other innovative products, such as the Watchman Left Atrial Appendage Closure Device and Alair Bronchial Thermoplasty System for the treatment of severe asthma, demonstrates our continued commitment to developing and bringing to market innovative products for physicians and their patients," Kucheman said in a prepared statement.
Mahoney is slated to take over as Boston Scientific president & CEO later this month.
The medical device titan is on a roll, having launched new heart and brain devices last week in Europe, MassHighTech reported.
Boston Scientific landed CE Mark approval in the European Union for its Vercise deep brain stimulation system, used in treatment of Parkinson’s disease, as well as European approval for the Emerge PTCA balloon catheter.

