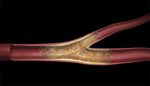
 Tryton Medical said today that it won pre-market approval from the FDA for its its side-branch stent for treating coronary bifurcation lesions and has already inked an exclusive U.S. distribution deal withCardinal Health (NYSE:CAH) subsidiary Cordis.
Tryton Medical said today that it won pre-market approval from the FDA for its its side-branch stent for treating coronary bifurcation lesions and has already inked an exclusive U.S. distribution deal withCardinal Health (NYSE:CAH) subsidiary Cordis.
The side-branch stent is designed to be compatible with any conventional drug-eluting stent in the main vessel, and comes in sizes ranging from 2.5mm to 3.5mm, Tryton said.
Although the Durham, N.C.-based company’s device failed to meet its primary endpoint for target vessel failure in a pivotal study with a target vessel failure rate of 17.4%, a subsequent analysis of patients with lesions suitable for a 2.5mm stent showed an 11.3% TVF rate, compared with 15.6% for the control cohort treating with provisional stenting.
“It is estimated that nearly a third of all patients treated with angioplasty each year have a bifurcation lesion, and the standard provisional treatment approach leads to side branch occlusion and bailout stenting far too often,” president & CEO Shawn McCarthy said in prepared remarks. “With this 1st-of-its-kind approval in the U.S., interventional cardiologists now have access to a stent that is specifically engineered to provide the complete lesion coverage and more predictable patient outcomes needed for the challenging anatomy of coronary bifurcation lesions.”
“Treatment of complex lesions at the site of a bifurcation has historically been inconsistent, with results varying depending on the procedure and the experience of the interventionist,” added CMO Dr. Aaron Kaplan. “A predictable bifurcation solution helps alleviate some of the stress in these procedures by limiting variability and reducing the need for bailout stenting. This important FDA decision could have a profound impact on treatment protocols and guidelines for significant bifurcation lesions in the years ahead.”
“This strategic agreement is a significant step for Cordis to quickly get innovative technologies into the hands of our customers in the U.S. We are actively preparing to commercially launch this product with Tryton to ensure physicians will soon have a new treatment option in their cath labs to help deliver the best patient care available,” Cordis president David Wilson said.

