An exclusive MassDevice investigation of 10 years of Food & Drug Administration data on its 510(k) approval process for medical devices shows that approval times for medical devices increased nearly 2 percent — while the number of 510(k) applications declined more than 19 percent — even as the FDA’s budgets and staff levels rose.
In fact, contrary to its oft-stated goal of clearing 90 percent of 510(k) applications in 90 days, the watchdog agency’s own data demonstrates that decision times are trending significantly longer. This and other key trends and data are available only in MassDevice.com’s Eye on the FDA report.
For example, the average decision time for 510(k) approvals was 113 days in 2008, compared with 87 days in 2005, an increase of 30 percent in just three years:
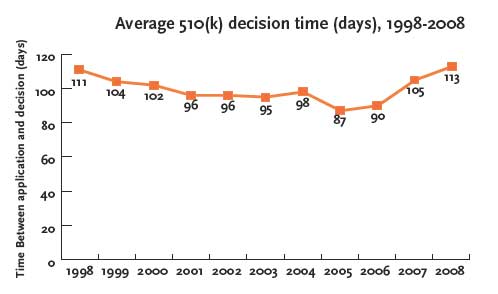
The report examines other key trends and data on the FDA’s 510(k) approvals process. Here’s a small sampling:
- Average decision times for 510(k) approvals
- Average decision times by device classification
- 10-year history of 510(k) approval decision times and device classifications
- A list of all the companies and devices that received 510(k) approvals during H1 2009
- The most active geographic regions for 510(k) approvals, by device classification
Hottest sectors and device classifications
“Medical device manufacturers should reconsider all timelines associated with the 510(k) approval process for medical technology,” said Brian Johnson, publisher of MassDevice.com. “Taking into account a new, more stringent approach on the part of the FDA’s new leadership and a comprehensive institutional examination of the 510(k) process that will likely result in recommendations for change, the regulatory path for new devices is likely to become longer, more costly and more onerous in the near future.”
Get the full report here.

