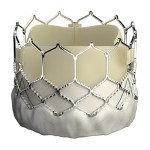
 Edwards Lifesciences (NYSE:EW) said today that it won CE Mark approval in the European Union for an expanded indication for its Sapien 3 transcatheter aortic valve replacement.
Edwards Lifesciences (NYSE:EW) said today that it won CE Mark approval in the European Union for an expanded indication for its Sapien 3 transcatheter aortic valve replacement.
The expanded indication covers patients at intermediate risk for open heart surgery, Irvine, Calif.-based Edwards said. Last month the FDA also expanded the Sapien 3 indication to include intermediate-risk aortic stenosis patients. The device won its original CE Mark approval back in January 2014, with the U.S. federal safety watchdog following suit in June 2015.
“This expanded intermediate-risk indication allows for the treatment of even more patients whose only previous option was an open-heart surgical procedure,” Dr. Helge Möllmann, of Dortmund, Germany’s St. Johannes Hospital, said in prepared remarks. “I am encouraged by the adoption of the position paper of the German Cardiac Society that recommends the use of transcatheter aortic valve implantation in intermediate-risk patients based on growing clinical evidence.”
Earlier today, Boston Scientific (NYSE:BSX) said it won CE Mark approval for its competing, next-generation Lotus Edge TAVR.

