French device maker EDAP (NSDQ:EDAP) announced last week that the FDA gave it a clean bill after inspecting its manufacturing facilities, putting the company one step closer to premarket approval for its ultrasound-based cancer therapy.
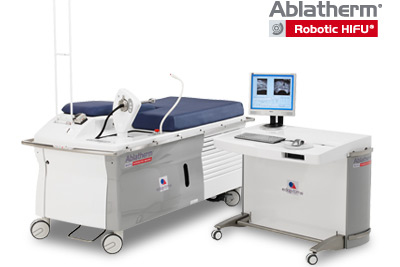
FDA inspectors found no issues and cited no observations after examining the facility where EDAP manufactures Ablatherm-HIFU, a system that uses high-intensity focused ultrasound for treatment of localized prostate cancer.
EDAP has been busy plowing its way through the FDA’s premarket approval process, raising funds and undergoing assessments ahead of its July date with the FDA. Earlier this month the company closed a $9.3 million funding round right on schedule, saying it plans to use the funds to prepare for its PMA meeting with the FDA’s Gastroenterology & Urology Devices Panel.
The clean inspection brings means EDAP can speed toward its FDA meetings on good terms, CEO Marc Oczachowski said.
"This is another important step in the FDA PMA process for our Ablatherm-HIFU technology," Oczachowski said in prepared remarks. "It confirms and validates EDAP’s ability to comply with high engineering, manufacturing and quality standards. In parallel, our team remains fully focused on preparing for the July 30, 2014 advisory committee meeting."
The minimally invasive Ablatherm platform uses high-intensity focused ultrasound to target tumors while leaving surrounding tissue intact. The device is already approved in the European Union, EDAP noted.
EDAP shares closed on Friday at $4.44, up more than 35% over the last month and more than 50% since the start of this year.

