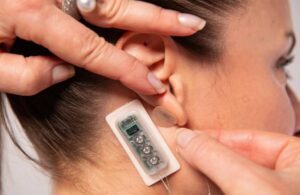
Palo Alto, California-based DyAnsys designed the wearable device, placed on the ear, to administer continuous pulses of low-level electrical current over several days, offering multiple treatments of up to 56 days for symptomatic relief of chronic, intractable pain from diabetic peripheral neuropathy.
“We are excited to have the FDA clearance of First Relief so that this device, which has been proven effective, can now be used to treat patients who have been experiencing pain related to diabetic neuropathy,” DyAnsys CEO Srini Nageshwar said in a news release. “First Relief offers a significant treatment option without drugs or narcotics.”
The FDA nod came after a study tested First Relief against a placebo and another previously FDA-cleared device. The study, conducted at the Jeevak Multispeciality Hospital in Warangal, India, evaluated 63 patients aged 30 to 74.
Devices were applied on a bi-weekly basis for 16 weeks with a primary efficacy endpoint of pain intensity measured through the Visual Analog Scale (VAS) score. Secondary efficacy endpoints were vibration perception threshold (VPT) value, insomnia severity index (ISI), overall neuropathy limitations scale (ONLS), Hamilton rating scale for anxiety.
The VAS pain score analysis demonstrated a significant reduction in the pain score of patients treated with First Relief, with the improvement persisting through the 90-day follow-up. Secondary outcome measures also showed similar improvements, while no complications or adverse events were observed during the study.

