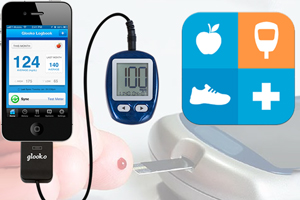
Silicon Valley mHealth company Glooko is expanding its FDA-approved mobile diabetes management system to include an app for the Android operating system.
Palo Alto, Calif.-based Glooko received 510(k) clearance from the FDA for the Android app and a new cable to connect to glucose monitors, a move company officials say will help it expand internationally.
Glooko’s diabetes management system is comprised of a $40 cable, available directly to patients, that connects with a smart phone. The system is unassuming in appearance, but its strength lies in its compatibility with a wide range of popular blood glucose testing systems. Glooko’s only prior clearance was only for Apple’s iPhone.
Join us to find out how Glooko will change chronic disease care on Dec. 10th
"Part of the special sauce inside of this cable is fairly complicated electronics that let us connect and communicate with lots of different meters and devices," Glooko CEO Rick Altinger told MassDevice.com earlier this year. "We have a couple of different kits that bring the data in, and then translate it into more of a standard format.
The company has partnered with a swath of diabetes device makers to gain access to their data specs, in some cases signing simple licensing agreements that grant them rights to the data and in other cases penning more in-depth deals that include sponsorship from the manufacturers. Those larger deals include a pact with Roche, 1 of the largest diabetes monitoring device makers in the world, as well as with WalMart for its ReliOn meter.
Altinger told Mobihealthnews that the expansion into the Android operating system enables the company to expand internationally.
“We’re highlighting the international numbers here, because Android has over 50% market penetration in the U.S., but in Europe it’s 65%,” he said. “So Android is especially important outside the U.S.”

