Go directly to MassDevice. Having trouble reading this email? View this newsletter in your browser here.
Not interested? Unsubscribe.
The MassDevice Weekly Roundup brings you the latest medical device news and information.
News
Deerfield Management to dump NeuroMetrix stake? ![]()
 Senate omits device tax amendment from healthcare reform bill
Senate omits device tax amendment from healthcare reform bill ![]()
Organogenesis files for PMA from the FDA for its CelTx dental tissue regeneration product ![]()
HeartWare boosts CEO Godshall’s pay ![]()
Hologic loses patent fight to SenoRx ![]()
Symmetry Medical to shutter Maine plant ![]()
Quest Diagnostics: Swine Flu declined 75 percent since October peak ![]()
Beckman Coulter confirms 2009 guidance ![]()
Medtronic introduces the Sovereign spinal system for lumbar surgery ![]()
Analyst: Medical device sector should outperform in 2010 ![]()
Caliper Life Sciences amends loan terms with Silicon Valley Bank ![]()
Thermo Fisher Scientific to spend $587 million in debt refinancing move ![]()
Abiomed touts another Impella study ![]()
AdvaMed launches diagnostics lobby ![]()
NxStage pays off again for Deerfield Capital ![]()
Interleukin launches bone health test ![]()
Brown looks to Green to keep National Dentex in the black ![]()
Covidien to ditch U.S. radiopharmacies ![]()
BioTrove spits out RapidFire spinout Biocious Life Sciences ![]()
Hocoma USA CEO Remsberg jumps ship for Tibion ![]()
![]()
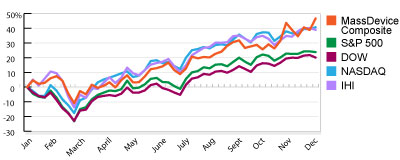
The MassDevice Checkup
Our weekly checkup takes the temperature of the public medical device companies in the three largest markets in the U.S.: Massachusetts, Minnesota and California. In addition to the indices for each state, which track the overall movement of the sector, we compare Monday morning’s opening share prices with their value when the markets close Friday afternoon and calculate the biggest gainers and losers. Read More ![]()
![]()
More News
Boston Scientific adds Tyco International exec to board ![]()
Thermo Fisher Scientific wins children’s health study contract ![]()
MTM Laboratories adds $10 million to Series C round ![]()
Helicos looks to scoop up $6.4 million ![]()
Thermo Fisher Scientific expands in N.C. ![]()
Robert Langer joins Millipore’s board ![]()
Athenahealth taps William Winkenwerder for board ![]()
Dirk Laukien sells off more of his Bruker stake ![]()
PatientKeeper rounds up $13 million ![]()
InVivo Therapeutics adds retired Siemens boss to board ![]()
BG Medicine, Corgenix ink biomarker supply deal ![]()
Phase Forward names new chief privacy officer ![]()
ProChon Biotech expands Biocart clinical trial ![]()
Federal judge dismisses shareholders’ lawsuit against NeuroMetrix ![]()
DePuy unit snaps up British orthopedic implant maker ![]()
Boston Scientific completes $2 billion bond offering ![]()
Eye on the FDA
MassDevice’s “Eye on the FDA” report examines key data on the FDA’s 510(k) clearance process on a semi-annual basis, analyzing the clearances according to medical discipline, clearance times and a variety of other categories. The reports characterize changes in the number of 510(k) clearances and decision times, identifying the most active medical disciplines and states and offering perspectives on clearance trends and developments in the regulatory landscape. Read More ![]()
![]()
Jobs
Featured Job: Product Manager (Burlington, Mass.)
Keystone Dental is looking for a dental implants product manager for a newly created position. The position entails taking products from concept to market, providing insights and analyses that drive growth and value. In addition, candidates will interface with the sales force, management and clinicians and be responsible for product market research activities, including demand and revenue forcasting and budgeting. Read More ![]()
![]()
Blogs
MassDevice blog: FDA: Mexico top exporter of medical devices to the U.S. last year
Mexico is the biggest exporter of medical devices to the U.S., according to the Food & Drug Administration. Read More ![]()
MedGadget’s MedTech Monday: Esteem totally implantable hearing device under consideration for approval in U.S.
A Food & Drug Administration committee considers approval of Envoy Medical’s fully implantable hearing aid; Bayer’s new blood glucose meter with USB connectivity; eXimia NBS system for non-invasive brain mapping gets U.S. OK; and a cardiopulmonary bypass pump brings attractive aesthetics to grim situations. Read More ![]()
MassDevice blog: Defibrillations caught on video
A pair of videos capture implantable cardiac defibrillators in action, as they kick in after a Danish climate scientist and a Belgian footballer experience irregular heart rhythms. Read More ![]()
Weekly Wireless Roundup: CardioNet hires financial advisor, mulls sale
CardioNet hired Lazard Freres & Co. of New York to evaluate its options; NYT, WSJ review Fitbit, DirectLife, Contour USB; Health devices with Bluetooth LE set for 2010; and the Center for Connected Health names its startup as SmartBeat. Read More ![]()
![]()
Upcoming Events
Featured Event
Jan. 6, Waltham, Mass. — Medical Development Group January Forum: Women’s Health: New Markets, Expanding Opportunities
“Learn about the latest advances is detecting and treating conditions that threaten their lives and the quality of those lives. From the latest methods of detecting and treating breast cancer to improving the lives of the sixteen percent of women experiencing incontinence, our clinical specialists and industry leaders will share their knowledge with you.”
Other events
Dec. 23, Boston — Massachusetts Life Sciences Center Board Meeting, 1 Ashburton, 21st floor, 3-5 p.m.
Want to list your event on our weekly newsletter? Contact us at editor@massdevice.com.
![]()
If you would like to subscribe to this newsletter, please visit our member registration page. ![]()
© 2009 Massachusetts Medical Devices Journal LLC and its licensors. All rights reserved. The material on this site may not be reproduced, distributed, transmitted, cached or otherwise used, except with the prior written permission of MMDJ.