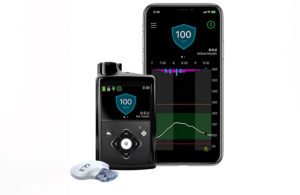
Results from real-world analyses in Europe and Chile, along with clinical data, were presented by Medtronic at the American Diabetes Association (ADA) 82nd Scientific Sessions in New Orleans.
According to a news release, the MiniMed 780G advanced hybrid closed loop system, designed with meal detection technology and automatic adjustments and corrections to glucose levels every five minutes, demonstrated improvements in glycemic outcomes that met consensus guideline recommendation of 70% time in range with less user interaction.
MiniMed 780G with the next-generation Guardian 4 sensor has not yet received FDA clearance, although Medtronic leadership anticipates the regulatory nod at some point during the company’s 2023 fiscal year.
Get the full story at our sister site, Drug Delivery Business News.

