Cordis, a Cardinal Health (NYSE:CAH) subsidiary, is recalling and removing certain lots of its Precise Pro RX carotid stent systems.
Cordis, a Cardinal Health (NYSE:CAH) subsidiary, is recalling and removing certain lots of its Precise Pro RX carotid stent systems.
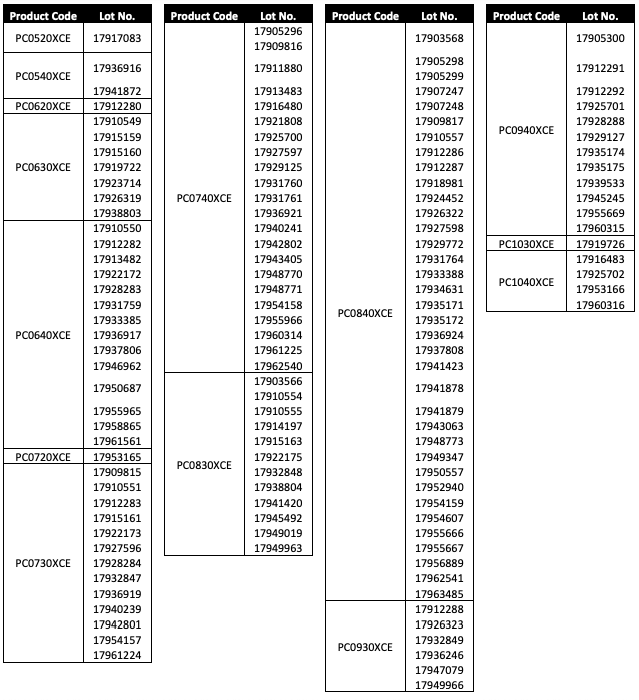
The Precise Pro RX carotid stent system, which is used in patients with stenotic lesions of the carotid arteries, has the potential for the distal tip to become separated from the wire lumen on certain lots of the system. The separation could result in an intra-procedural delay while a replacement device is prepared, unplanned percutaneous or surgical intervention or stroke, according to an urgent field safety notice.
Santa Clara, Calif.-based Cordis said it recently confirmed complaints about distal tip separation may be the result of inadequate joint adhesion. There have been no reports of strokes, deaths or other long-term patient events related to the distal tip separation.
Cordis is recommending healthcare providers check inventories to confirm if there are any units from the affected lots and to return the devices to the Cardinal Health distribution center.
Devices involved in the recall were made between October 2019 and August 2020.