
Contura Inc.’s pre-market approval application for its Aquamid dermal filler is slated for an appearance before an FDA advisory panel in December.
The product is a permanent, “soft volume” injection designed to fill the voids underneath the surface of the skin that cause facial wrinkles. It’s biocompatible and non-allergenic; Soeborg, Denmark-based Contura claims that “its high water content makes the treated area look and feel incredibly soft and natural,” according to its website.
The PMA application for Aquamid will go before the watchdog agency’s General and Plastic Surgery Devices panel Dec. 1 for a public hearing. Recommendations from its advisory panels aren’t binding on the FDA, but it usually goes along with their votes.
Sign up to get our free newsletters delivered straight to your inbox
The Contura PMA discussion will likely cover the peer-reviewed data from clinical trials involving more than 5,000 subjects. Another likely topic is the safety risks associated with Aquamid, which “were mostly mild transient local reactions,” according to the website.
“In rare cases, local infections have occurred and have been resolved with antibiotic treatment,” according to the site.
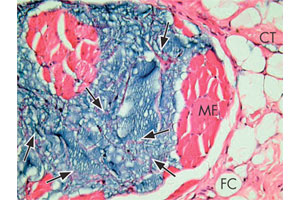
Microphoto of Aquamid (see arrows) injected into the chin four years previously. It is surrounded by muscle fibres (MF), fat cells (FC) and connective tissue (CT). It is anchored by a fine network of host connective tissue strands (arrows).
Source: Contura Inc.

