Australian-based Cochlear Ltd. (ASX:COH), the world’s biggest maker of bionic ears, saw stocks drop by nearly 20 percent after a voluntary recall of its popular titanium hearing implants.
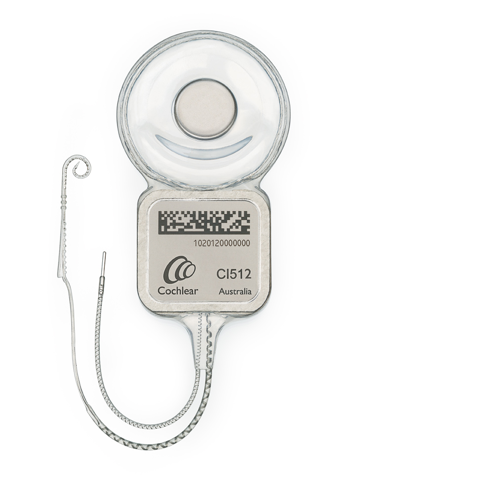
Cochlear recalled its Nucleus CI500 range of implants from shelves after an unexplained increase in failures in its CI512 implant model.
The recall affects sales in 27 countries, including the U.S. and Australia. Shares dropped 20 percent since the announcement today, closing at $57.50 from yesterday’s close at $72.18.
The CI500 made up 70 percent of the company’s sales of implant revenue and about half of Cochlear’s annual sales of more than $800 million, MarketWatch reported.
According to Cochlear, there are about 25,000 registered users of the CI500 range and the company isn’t recalling those products that have already been implanted. Less than one percent of the CI512 implants have failed since launched in 2009.
If a failure occurs, the implant safely shuts down without injuring the patient, company representatives said in a prepared statement. Cochlear representitives added that if its device does fail, patients can have their implant replaced with an earlier model from Cochlear’s Nucleus Freedom range, which were not affected by the recall.
"Reliability is very important," CEO Chris Roberts told analysts on a conference call. "If there’s some things happening that we don’t understand, we’re far, far better to stop and understand it…rather than just battle ahead and hope it all works out."
The Nucleus CI500 implant range includes the Nucleus CI512, Nucleus CI513, Nucleus CI551 double array implant and Nucleus ABI 541 Auditory Brainstem Implant.

