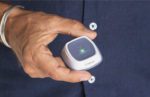
Coala Life announced today that it received FDA emergency use authorization (EUA) for an expanded indication for its Coala Heart Monitor.
The expanded indication for the heart monitor enables physicians to remotely monitor heart and lung sounds in real-time, which can be used in COVID-19 respiratory diagnostic exams. Coala, a Swedish company with a location in Irvine, Calif., introduced the smartphone-powered monitor in Scandinavia in 2017.
With the Coala Heart Monitor, patients who are quarantined as a result of the COVID-19 pandemic can monitor their heart and record their lung sounds in 60 seconds while physicians remotely read the analyzed data, according to a news release. Physicians also have access to the patient’s electrocardiogram (ECG) and can listen to respiratory sounds from a remote location.
The Coala Heart Monitor does not require hospital-based enrollment and is capable of activation at home, eliminating any need for patches or wires. Coala said in the release that demand for its heart monitors is already high in Europe among high-risk patients with coronary and pulmonary diseases, as well as those of advanced age.
“The Coala device was developed and validated in Sweden based on over ten years of research,” Coala Life founder and president Philip Siberg said in the release. “It truly enables telemedicine with the ability to monitor respiratory sounds and cardiac-related conditions in the safety of patient’s homes and help eliminate the need for patient office visits.”

