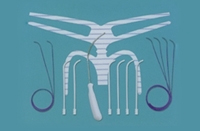
New details on Johnson & Johnson’s (NYSE:JNJ) marketing of its controversial transvaginal mesh implants are again in the spotlight as unsealed documents suggest that the healthcare giant sold the devices for an additional 9 months after the FDA deemed the products adulterated and asked J&J to pull them off the shelves.










