
A California jury ruled against Johnson & Johnson (NYSE:JNJ) and its DePuy subsidiary in finding that the company’s ASR XL metal-on-metal hip implant was defectively designed.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
FDA | Recalls | 510(k) | Pre-Market Approvals | MDSAP | Clinical Trials

A California jury ruled against Johnson & Johnson (NYSE:JNJ) and its DePuy subsidiary in finding that the company’s ASR XL metal-on-metal hip implant was defectively designed.

LifeScan, has received regulatory clearance from the FDA for its OneTouch VerioSync blood glucose monitoring system, the first monitoring system to integrate with the Apple app store.
The app, called the OneTouch Reveal, is available on iPhone, iPad or iPod touch.
The system automatically sends blood glucose levels to mobile devices, displaying information in a 14-day summary with simple visual breakdowns. Patients can also share information via text or email or with their doctors or friends.

MASSDEVICE ON CALL — Federal healthcare regulators gave Ad-Tech Medical Instrument Corp.’s wide-reaching brain implant recall Class I status, the highest-risk category for medical device recalls.
Late last year the Wisconsin-based medical device maker recalled its Macro Micro Subdural Electrodes over concerns that the implanted devices could cause damage to the brain.
Welcome to the latest edition of the MassDevice.com’s D+Vice Talk, our new webcast, in which we review medical device stories from the last week.
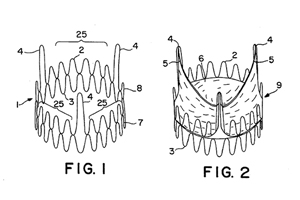
Drawings from Edwards Lifesciences’ U.S. Patent No. 5,411,552.


The FDA issued new guidance governing premarket notification submissions for pulse oximeters. The new guidelines apply to all 510(k) submissions for the non-invasive blood oxygen level and pulse rate measuring devices.

Surgical devices maker ArthroCare (NSDQ:ARTC) won FDA clearance for its SpeedLock HIP implant, a soft-tissue-to-bone fixation implant for hip repair surgeries.
The device’s primary use is in acetabular labral reattachment and hip capsular repair, according to a press release.

By: Bakul Patel, MS, MBA
Calling all movers and shakers in health care information technology!
We’re on a mission to help pave the way for innovative advances in safe and effective health information technology (HIT).
Who are "we"?

