
Technology titan Apple Inc. (NSDQ:AAPL) sent a few executives to the FDA late last year to meet with agency officials to talk about "mobile medical applications," according to the FDA’s public calendar.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Technology titan Apple Inc. (NSDQ:AAPL) sent a few executives to the FDA late last year to meet with agency officials to talk about "mobile medical applications," according to the FDA’s public calendar.

By Stewart Eisenhart, Emergo Group
The US Food and Drug Administration has released two new draft guidances for medical device manufacturers that either include computational modeling data in their registration applications or use animal-derived sources in their products.

MASSDEVICE ON CALL — The FDA this month issued a formal warning to the Institutional Review Board of Mercy Hospital & Medical Center, citing the committee for violations of policies regarding human clinical testing.
The feds noted record-keeping and protocol violations regarding the board’s clinical trial , the keeping of meeting minutes and other breaches, and said that the board’s initial responses to some of the concerns were "inadequate."

MASSDEVICE ON CALL — Based on the results of a newly unveiled study, the FDA plans to implement new processes to ensure that women make up an appropriate portion of medical device post-approval studies.
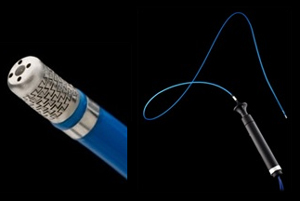
Medtech titan St. Jude Medical (NYSE:STJ) won FDA premarket approval for its Cool Flex cardiac ablation system with indication to treat irregular heart rhythms.

The U.S. Senate yesterday approved an omnibus, $1.012 trillion budget bill that includes the restoration of some $85 million in user fees from medical device and pharmaceutical companies that was held in abeyance by sequestration.
President Barack Obama, who has until midnight tomorrow to sign the measure into law, is expected to quickly add his signature after the 72-26 vote in the Upper Chamber.

By: Margaret A. Hamburg, M.D.
Ensuring the safety of the food supply and the safety and effectiveness of drugs, devices and biologics has always been at the core of FDA’s responsibility to protect the public health – and always will be.

