
Class I recalls
Recalling Manufacturer, Date Posted, Trade Name/Product, Reason for Recall
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

Recalling Manufacturer, Date Posted, Trade Name/Product, Reason for Recall
Updated August 30, 2013, at 4:00 p.m. with comments from Stryker.

The FDA issued a Class I recall for 5 lots of Stryker Spine’s Oasys Midline Occipital Plate, warning patients and physicians that the device might fracture after surgery.

Medical device makers issued more than 400 recalls during Q2, 30% more than in the previous quarter. That’s higher than reported for the last 5 quarter, according to a report by Stericycle ExpertRECALL
The recalls affected about 9.2 million units, a 50% decrease in the quantity of products affected in Q1. Four recalls affected between 500,000 and 1 million units each, and no single recall affected more than 1 million products.

After Nova Biomedical issued a voluntary recall letter for its Nova Max test strips and glucose meter kits, the FDA determined that the measure qualified as a Class I recall, a label reserved for issues that pose a threat of significant patient harm.

Regulators in the UK and Hong Kong issued warnings on CircuLite’s Synergy circulatory support system after at least 3 reports that the devices’ inflow cannulae fractured.
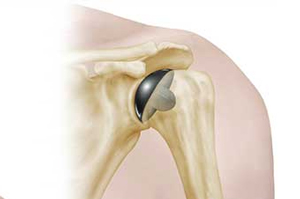
In consultation with Australian health authorities, LMT Surgical issued a warning to all surgeons who implanted the company’s PyroTitan shoulder implant, advising of a reported 3% failure rate in the devices.

Medical device company Aimedics is pulling all units of its HypoMon sleep monitoring device off the shelves in Australia following reports that the device’s alarm does not work as well as stated.
HypoMon is a night-time device for children and young adults with Type 1 diabetes, designed to sound an alarm if the patient’s blood-sugar level drops too low while sleeping.

Florida-based infusion device company Iradimed landed in hot water with some units of its MRidium infusion pumps, the FDA giving the measure it’s highest-risk Class I recall status, a label reserved for the most serious device malfunctions that might result in death.

The FDA sent out the highest-risk recall notice for the an infusion pump made by CareFusion (NYSE:CFN), the most recent warning to customers involving the company’s Alaris product line.

