
Reps. Mike Rogers (R-Mich.) and Ed Markey (D-Mass.)
Congressional Representatives Ed Markey (D-Mass.) and Mike Rogers (R-Mich.) introduced a bill that would require medical device makers to include pediatric information in certain FDA applications.
The same rule had been earlier withdrawn by the FDA after the agency received "significant adverse comment" upon implementing the change in the summer of 2010.






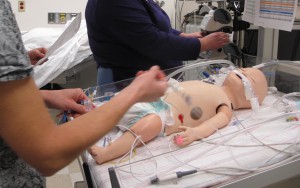 Medical simulators give doctors and nurses an opportunity to practice without risking patient harm. Peter Weinstock and his team are creating sophisticated, scenario-based simulations that bring new realism to medical training.
Medical simulators give doctors and nurses an opportunity to practice without risking patient harm. Peter Weinstock and his team are creating sophisticated, scenario-based simulations that bring new realism to medical training.

