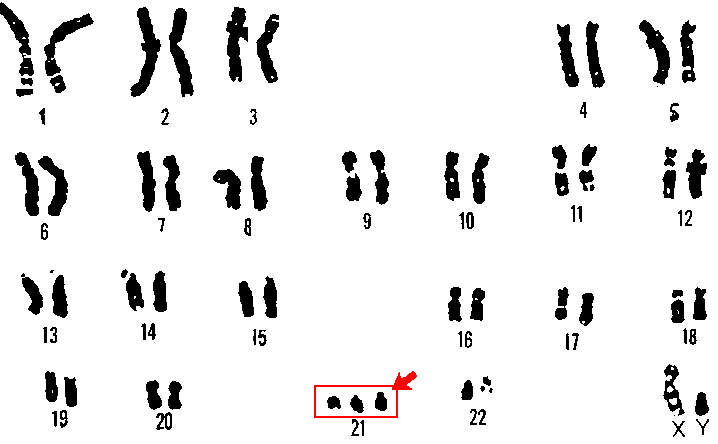
Researchers have silenced the third copy of chromosome 21, at least in a dish. What might this mean for Down syndrome? (Wikimedia Commons)
The Medical Device Business Journal — Medical Device News & Articles | MassDevice


By Tom Ulrich
This morning the Wall Street Journal’s Ron Winslow published a story, “Advances that Regrow Babies’ Hearts,” featuring work by Boston Children’s Hospital cardiac surgeon Sitaram Emani, MD, on new ways of treating hypoplastic left heart syndrome (HLHS).

One of the greatest pleasures I have as FDA commissioner is the opportunity to meet with so many who are making a real difference in the world of health care, including researchers, doctors, drug industry leaders, foundations, and advocates for patients and families, including most recently the Alexandria Summit for Oncology.

A slippery coating inspired by the surface of a pitcher plant could help keep IV lines free of bacteria and blood clots. (kleo_marlo/Flickr)
By Tom Ulrich
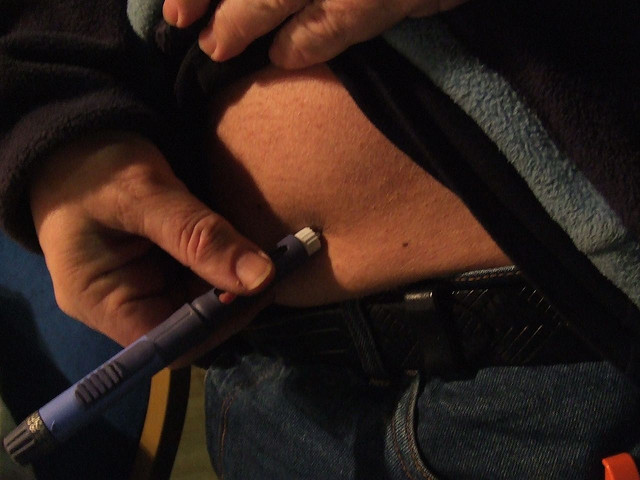
By Tripp Underwood
For decades, patients have managed their type 1 diabetes by injecting themselves with insulin to regulate the glucose in their blood. While this form of medical management addresses the immediate danger of low insulin levels, long-term complications associated with diabetes, like heart and kidney diseases, still threaten more than 215,000 children currently living with the disease in the United States.

By Tom Ulrich


Reducing health care costs doesn’t have to involve making sacrifices in patient safety or quality of care or holding clinicians to rigid guidelines. Over the past several years, Boston Children’s Hospital has rolled out a methodology known as Standardized Clinical Assessment and Management Plans (SCAMPs).


Advances in medical care sometimes present challenges on the flipside. Case in point: Over the past three decades, progressive developments in pediatric cardiac care have allowed many babies born with congenital heart disease (CHD) to survive. And longevity continues to improve. This progress, however, has brought hospitals a burgeoning patient population with tremendously complex and varied disease states.

