
MASSDEVICE ON CALL — Johnson & Johnson (NYSE:JNJ) and its Ethicon subsidiary notched their 1st lawsuit since the company suspended sales of its laparoscopic power morcellators.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

MASSDEVICE ON CALL — Johnson & Johnson (NYSE:JNJ) and its Ethicon subsidiary notched their 1st lawsuit since the company suspended sales of its laparoscopic power morcellators.

Medtech titan Boston Scientific (NYSE:BSX) announced today that it acquired IoGyn, maker of the Symphion system for removing intrauterine tissue, for $65 million, not including previous investments.

Endo Health Solutions (NSDQ:ENDP) said it struck an $830 million deal to settle thousands of lawsuits over its American Medical Systems pelvic mesh products.
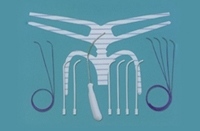
The FDA today issued a pair of proposed orders that would increase regulatory oversight of controversial pelvic mesh products, responding to a long-running public uproar about the implants’ potential for harm to women.
Johnson & Johnson‘s (NYSE:JNJ) Ethicon subsidiary asked a federal judge yesterday to block a bid to send a group of pelvic mesh product liability cases back to a state court in Philadelphia.

Endo Health Solutions (NSDQ:ENDP) said the FDA warned it about procedural violations at a Minnesota plant run by its American Medical Systems subsidiary that makes pelvic mesh products.

MASSDEVICE ON CALL — The Corporate Action Network is looking for tips to help take down healthcare giant Johnson & Johnson (NYSE:JNJ) and its CEO, Alex Gorsky.
A Dallas jury split its decision in a product liability lawsuit filed over Ethicon’s TVT-O pelvic mesh yesterday, awarding $1.2 million in compensatory damages to plaintiff Linda Batiste.


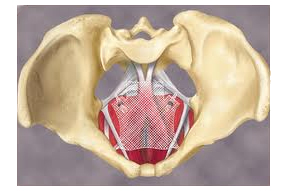
The European Commission asked a scientific advisory panel to look into the safety of surgical procedures to treat urogynecological conditions using mesh products, which have embroiled their makers in tens of thousands of product liability lawsuits in the U.S.
The EU’s executive body asked its Scientific Committee on Emerging and Newly Identified Health Risks for an opinion on the "safety of surgical meshes used in urogynecological surgery," according to the commission’s request.

