Food & Drug Administration (FDA)
FDA OKs Covidien’s Nellcor portable oxygen monitor
FDA panel recommends PMA process for CryoLife heart valve
FDA approves Bard’s Lutonix DEB
FDA and the cybersecurity community: Working together to protect the public health

By Suzanne Schwartz, M.D., M.B.A.
Cyber vulnerabilities – bugs or loopholes in software codes or other unintentional access points – are a real and constant threat to our networked laptops, mobile phones, or tablets. The Heartbleed virus and security breaches at major retailers are just a few recent examples of exploits of this hazard that have been in the news.
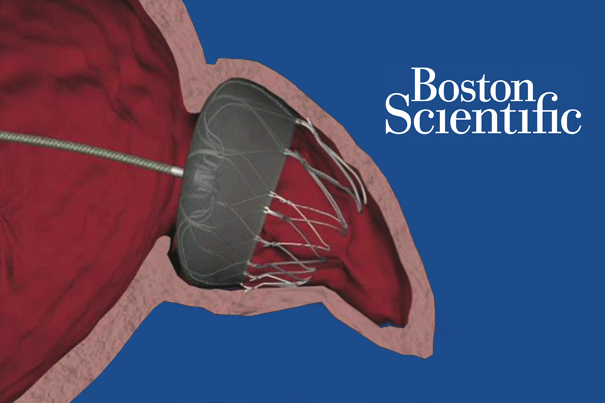
UPDATE: 3rd FDA panel vote barely favors Boston Scientific’s Watchman anti-stroke device
FDA’s program alignment addresses new regulatory challenges

By Margaret A. Hamburg, M.D.
Over the last year, a group of senior FDA leaders, under my direction, were tasked to develop plans to modify FDA’s functions and processes in order to address new regulatory challenges. Among these challenges are: the increasing breadth and complexity of FDA’s mandate; the impact of globalization on the food and medical product supply chains; and the ongoing trend of rapid scientific innovation and increased biomedical discovery.