
The FDA granted Direct Flow Medical an investigational device exemption for its transcatheter aortic valve implant, with the medical device company announcing enrollment in its Salus feasibility trial.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

The FDA granted Direct Flow Medical an investigational device exemption for its transcatheter aortic valve implant, with the medical device company announcing enrollment in its Salus feasibility trial.

St. Jude Medical (NYSE:STJ) today announced CE Mark approval of its Ilumien Optis PCI optimization system, designed to help physicians assess and treat patients with coronary artery disease.

Boston Scientific (NYSE:BSX) shares hit a new 52-week high today after the medical device company announced the launch of a clinical trial of its WallFlex biliary stent.
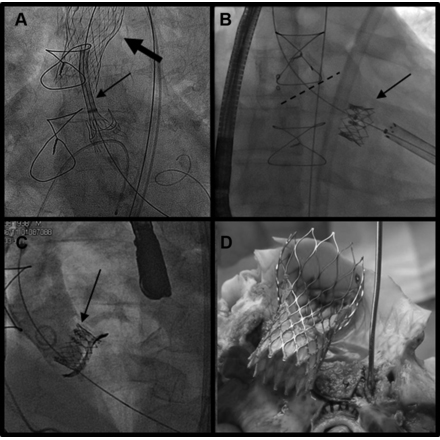
The short-term results of minimally invasive transcatheter valve-in-valve implantation to treat failed biological heart valve protheses are "excellent," but patients must be closely monitored due to some notable cases of suboptimal systolic-valve performance, according to early data from a multi-center trial.

Edwards Lifesciences (NYSE:EW) said it plans to buy back another $750 million worth of its own stock during the 2nd quarter, with CEO Michael Mussallem buying another $5 million worth on his own.


With a green-light from the FDA, Elixir Medical Corporation will move forward with its Excella III pivotal trial to review its DESyne Nx Novolimus-eluting coronary stent system. Elixir plans to enroll patients at 50 clinical locations and will use the trial results to support its upcoming premarket approval application.

St. Jude Medical (NYSE:STJ) won European regulatory approval for 2 next-generation defibrillators, the Ellipse and the SJM Assura high voltage systems. The new devices aim to bolster device reliability by preventing lead-to-can abrasion, according to the company.

When the New York Post broke the story this week that New Jersey governor and presidential hopeful Chris Christie had underwent weight loss surgery using the LAP-BAND in February some on Wall Street bemoaned the rotund politicians timing.


OrbusNeich Medical wasted no time in enforcing a patent win against Boston Scientific (NYSE:BSX), announcing that it is blocking German imports of its rival’s cardiac stents.

