
San Diego medical device startup Acutus Medical closed its Series B funding round with $28 million raised from a handful of investors in support of the company’s minimally invasive heart mapping technology.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
 Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
🫀 Narrow your focus with these categories:
Cardiac Implants | Structural Heart | Pulsed-Field Ablation
Here are top cardiology features from MassDevice and its sister publications (and scroll down to read the latest cardiology device news from MassDevice):
+ LVADs save lives: So why aren’t more available?
+ How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
+ How Abbott designed the world’s first dual-chamber leadless pacemaker system
+ J&J used RWE for expanded indications — and you can, too
+ 7 cardiology startups you need to know

San Diego medical device startup Acutus Medical closed its Series B funding round with $28 million raised from a handful of investors in support of the company’s minimally invasive heart mapping technology.

A month after regulatory approval of the Sapien XT heart valve in Japan, Edwards Lifesciences (NYSE:EW) said it won reimbursement for the transcatheter aortic valve implant from Japan’s Central Social Insurance Medical Council.

A pair of Canadian women, aged 87 and 71, became the 1st non-European patients to receive treatment with JenaValve Technology‘s new transcatheter aortic valve implantation system.
JenaValve’s 2nd-generation transapical TAVI device is surgically implanted in the natural aortic valve cavity, and features an anchoring and clipping mechanism to ensure the device stays in place.
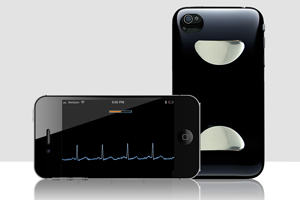
AliveCor’s iPhone-based electrocardiogram monitor bears all the hallmarks of modern mobile health technology, but the cheap, hand-held device also facilitates routine, clinic-based screening without the aid of a specialist. In studies taking place Down Under, some clinical receptionists screen patients for heart rhythm disorders before the patients ever sees the doctor.
Patients with severe mitral regurgitation showed significantly better survival rates after early surgery, compared with a "watchful waiting" approach, according to a study published this week in the Journal of the American Medical Assn.

Regulators in the UK and Hong Kong issued warnings on CircuLite’s Synergy circulatory support system after at least 3 reports that the devices’ inflow cannulae fractured.

Medical device giant Boston Scientific (NYSE:BSX) listed no fewer than a dozen legal battles in its latest financial report, notching some wins, some losses and some seemingly circuitous fights without end.

Premier Healthcare Alliance awarded several new contracts for drug-eluting stents, traditional stents and other intervention products to 8 heavy hitters in the cardiology space.

MASSDEVICE ON CALL — To treat aortoiliac occlusive disease, a narrowing of the aorta or iliac arteries, cardiologists typically must choose between open bypass surgery or a less-invasive endovascular treatment.
A new meta-analysis shed some light on the pros and cons of each procedure, concluding open bypass is the more durable option but that it comes along with a longer hospital stay and increased risk of complications and death.

Xlumena closed a $4.8 million debt and securities round with 10 unnamed investors.
Founded in 2007, the Mountain View, Calif.-based company makes ultrasound-guided interventional microscopes, including an FDA-cleared pancreatic cyst access device called Navix.
Xlumena is wrapping up clinical trials to support a 510(k) application for its Axios stent and delivery system, also to treat pancreatic cysts.

