Cardiovascular
 Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
🫀 Narrow your focus with these categories:
Cardiac Implants | Structural Heart | Pulsed-Field Ablation
Here are top cardiology features from MassDevice and its sister publications (and scroll down to read the latest cardiology device news from MassDevice):
+ LVADs save lives: So why aren’t more available?
+ How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
+ How Abbott designed the world’s first dual-chamber leadless pacemaker system
+ J&J used RWE for expanded indications — and you can, too
+ 7 cardiology startups you need to know
Latest Cardiology News
Maquet inks marketing deals with ClearFlow, InterValve
Infraredx sets $56m IPO terms and hits the road
Infraredx today set the terms for its forthcoming initial public offering, saying it plans to offer 4 million shares at a range of $13 to $15 each, and set out on a road show to drum up interest from investors.
Boston Scientific hits 7-year high
Kips Bay Medical slashes workforce
SynCardia wins FDA nod for destination therapy trial
SynCardia plans to test its artificial heart as a permanent implant rather than a placeholder for donor transplants in 19 patients. The Arizona company said it won the FDA’s approval to move ahead with the clinical study of its temporary Total Artificial Heart as a permanent transplant option. This is a big advance forward for […]
Philips agrees to pay $1B for Volcano
Infraredx study: NIRS may help assess cardiac risk
Infraredx touted a study in the Journal of the American College of Cardiology showing its imaging technology could help cardiologists assess a patient’s risk for suffering a heart attack or stroke.
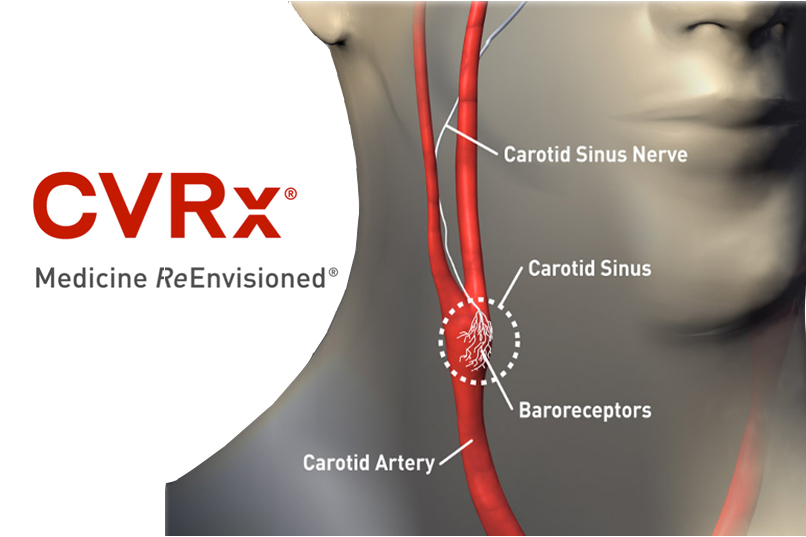
CVRx’s Barostim neo gets CE Mark for use with MRIs
CVRx said its Barostim neo system was granted expanded CE Mark approval in the European Union for use with MRI procedures.
TAVI: Transcatheter Technologies to develop transfemoral valve
Edwards CEO Mussallem hopes FDA to accelerate approvals of medical devices

(Reuters) – U.S. health regulators appear open to speeding up approvals of medical devices that currently often reach European markets three to five years ahead of being cleared in the United States, the chief executive officer of Edwards Lifesciences (NYSE:EW) said.