Blog
Research registry reveals new mutations in rare childhood blood disorders

By Tom Ulrich
To really understand rare conditions, you need a lot of data from a lot of patients. But no one hospital or center usually sees more than a few patients with any given rare disease, precisely because they’re rare.
It’s hip to be social

By Rob Kinslow, Sr. Strategist, Brand Communication, KHJ Brand Activation
CEO Insight: Allergan’s David Pyott on demonstrating value

By David Pyott
A simple idea for collaborative academic research

With the rapid dissemination of medical information, global reach of the internet, and realization that more print journals doesn’t mean better print journals, today’s researchers use a variety of techniques to connect but most, sadly, have not kept pace with the times.
Working together for new solutions to cancer in children

One of the greatest pleasures I have as FDA commissioner is the opportunity to meet with so many who are making a real difference in the world of health care, including researchers, doctors, drug industry leaders, foundations, and advocates for patients and families, including most recently the Alexandria Summit for Oncology.
New rules for supplementary information requests in China

By Stewart Eisenhart, Emergo Group
Chinese medical device market regulators have enacted a new requirement regarding supplementary documentation requests (links to page in Chinese) related to registration with the China Food and Drug Administration (CFDA).
Effective June 1 2013, medical device registration applicants that receive CFDA requests for supplementary information must comply with the following rules:
Hot enough for you? Keeping babies warm in developing countries
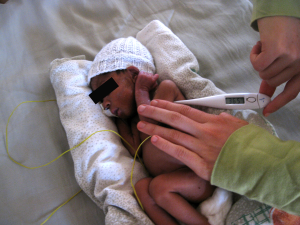
Newborns like this child have a high risk of hypothermia, even in warm climates. An innovative warming pad could be one potential fix. (Courtesy of Anne Hansen)
It’s a mad mad mad mad medtech world

Reuters recently reported that a growing number of U.S. cardiologists are expressing frustration with the lack of availability of newer cardiac devices in the United States as compared to Europe. Their beef: U.S. patients have to wait longer for access to beneficial next generation therapies.
Changing the game, one rule at a time
Is a merger or an acquisition rocking your world? As you grapple with how to integrate products, process and performance, don’t forget about your people. Employees are your culture, your reputation, your brand in action. In fact, companies that invest in their internal brand show more than 50% greater ROI in many areas, including employee engagement, retention, productivity and customer satisfaction.
Join us for “Changing the game, one rule at a time,” a conversation about how to turn marketing inside out.
Speakers:
South Korea revises medical device regulations

By Stewart Eisenhart, Emergo Group
South Korea’s Ministry of Food and Drug Safety (MFDS) has made three significant amendments to the country’s Medical Devices Act that will affect some market registrants going forward.


