Boston Scientific aims for double-digit earnings growth
May 1, 2015 by Brad Perriello
Boston Scientific CEO Mike Mahoney, who’s overseen a turnaround at the medical device giant since taking over the corner office in 2012, today said the company is targeting double-digit earnings growth each year for the next 6 years.
Mahoney, speaking at the Marlborough, Mass.-based company’s investor day presentation in New York this morning, said Boston expects to post adjusted earnings per share of 88¢ to 92¢ this year, excluding foreign exchange effects. Read more
![]()
![]()
Profound Medical goes public in reverse merger
May 1, 2015 by Mark Hollmer
Profound Medical said it’s slated to hit the public markets in Canada after a reverse merger, also revealing a $24 million private placement to fuel clinical trial work.
The proceeds will help support Profound Medical’s 110-patient pivotal trial of the Tulsa-Pro MRI-guided ultrasound ablation device to treat prostate cancer. Read more
![]()
Pavmed files for $23m IPO
May 4, 2015 by Brad Perriello
The entrepreneurs behind a quartet of medical device companies is betting their formula can cut down on the time and cost of getting medtech to market with a $23 million initial public offering.
Pavmed is developing devices in 5 areas, according to its IPO registration: The PortIO long-term implantable vascular access device; the Caldus line disposable tissue ablation devices, including renal denervation for hypertension; the CarpX percutaneous device to treat carpal tunnel syndrome; the NextCath self-anchoring short-term catheter; and the NextFlo disposable infusion pump. Read more
![]()
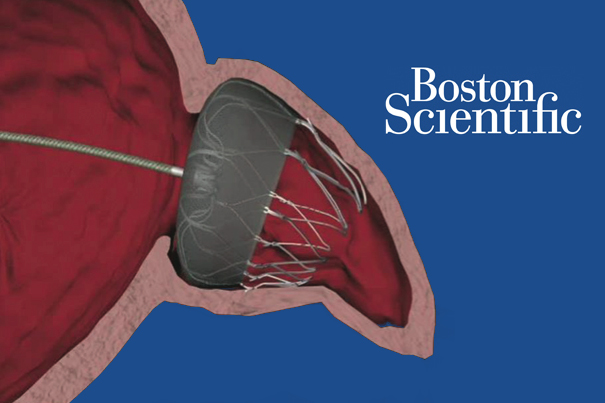
Boston Scientific's Mahoney is bullish on Watchman
April 29, 2015 by MassDevice
Boston Scientific expects more hospitals will be able to offer its new atrial fibrillation treatment starting by the end of the 3rd quarter as it paces the product’s rollout to give physicians time to learn how to use it, CEO Mike Mahoney said yesterday.
The device, called Watchman, was approved by U.S. regulators last month as an alternative to long-term use of blood thinners such as warfarin for patients at high risk of stroke. Read more
![]()
Euronext suspends Carmat shares after artificial heart patient dies
May 5, 2015 by MassDevice
Trading in Carmat shares was suspended after the company’s 2nd artificial heart patient died May 2, 9 months after receiving the transplant.
A source close to the matter said the Euronext stock exchange granted the suspension to give medical teams time to pinpoint the reasons for the death, and that a statement could be issued after the market’s close. Read more