The medical device giant plans to start offering the Micra AV in a limited number of implanting centers in the coming weeks, with a full launch expected in the spring.
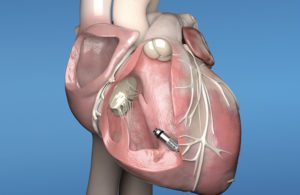
FDA initially approved the Micra system in 2016. It’s about the size of a vitamin pill. Implanted inside the heart’s right ventricle via catheter, it does away with the need to run leads to the heart. The system, though, hasn’t been able to provide the therapies associated with dual-chamber pacing systems — until now.
The new Micra AV has the same size and shape as the original Micra system, but it has several additional internal atrial sensing algorithms that detect cardiac movement. The Micra AV is able to adjust pacing in the ventricle to coordinate with the atrium, enabling it to provide “AV synchronous” pacing therapy to people with atrioventricular block. People with AV block have previously needed a dual-chamber pacemaker, even though only one of the two leads in system provided heart stimulation.
The new indication for people with AV block means that leadless pacemakers are now an option for half of the people requiring pacemakers, Medtronic spokesman Ryan Mathre said when reached by MassDevice. The FDA approval marks an expansion of what is arguably one of the top medtech innovations of the past decade.
“With the approval of Micra AV, more pacemaker patients qualify for a new treatment option that offers the advantages of leadless pacing – including a minimally invasive implant procedure and a cosmetically invisible device,” said Dr. Larry Chinitz, cardiac electrophysiologist and director of NYU Langone’s Heart Rhythm Center in New York City.
“Although complications with traditional pacemakers are infrequent, when they occur, they’re expensive to treat and can be invasive for the patient. Real-world use of Micra has shown a 63% reduction in major complications compared to traditional pacemakers,” Chinitz said in a news release.
MDT stock was up more than 1%, to $120.32 per share, by the close of trading today.

