Boston Scientific (NYSE:BSX) yesterday revealed a trifecta of reimbursement wins for its S-ICD subcutaneous defibrillator from the American Medical Assn., Medicare and Aetna.
Marlborough, Mass.-based Boston Scientific said the AMA created new reimbursement codes for the S-ICD, slated to go into effect Jan. 1, 2015. That’s also when a 23% boost to the Medicare hospital outpatient payment rate for S-ICD implantation is scheduled to kick in, according to a press release.
Boston Scientific also said that Aetna broadened its coverage for the device to ICD-eligible patients.
"These important milestones will facilitate payment processing for both physicians and hospitals," chief medical officer for rhythm management Dr. Kenneth Stein said in prepared remarks. "Along with the favorable published safety and efficacy data in peer-reviewed journals, these code and payment changes will further enable rapid acceptance of this ground-breaking therapy by the medical community.”
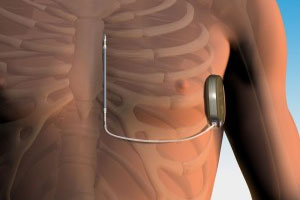
The S-ICD differs from traditional ICDs in that the leads that transmit electricity to the heart are implanted in a surgically created channel along the patient’s chest; the regulating shock is delivered to the heart through the breastbone.
"The S-ICD System is an advanced, less invasive option for patients at risk of sudden cardiac arrest," Dr. Michael Gold of the Medical University of South Carolina said in a statement. "The new CPT categorization establishes more predictable coding processes for hospitals and physicians when implanting the S-ICD System. This change should improve patient access to this important therapy."