Boston Scientific (NYSE:BSX) kicked off a new registry evaluating deep brain stimulation in treatment of Parkinson’s disease.
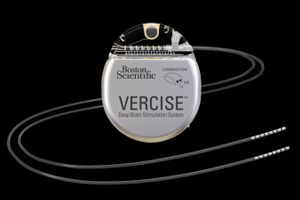
The medtech titan announced this week that it enrolled the 1st of up to 300 patients slated for participation in the registry. Researchers will follow patients implanted with Boston Scientific’s Vercise DBS system, an implantable neurostimulator that selectively stimulates targeted areas of the brain associated with movement disorders.
The registry adds to ongoing studies of the Vercise implant, including a U.S. clinical trial launched just last year.
"This registry will enable us to provide physicians with a comprehensive, long term view of real world experience and results with the Vercise DBS System for the many patients affected by Parkinson’s disease," neuromodulation president Maulik Nanavaty said in prepared remarks.
The Vercise technology already has regulatory approval from healthcare officials in the European Union and Australia and is on the market in Europe, Israel, Australia and select countries in Latin America. The device also has European CE Mark approval for treatment of primary and secondary dystonia.
The device has only investigational approval from the FDA and is not available for sale in the U.S.

