BaroNova said today that it won FDA approval for its TransPyloric Shuttle non-surgical device intended for treating obesity, now cleared as a weight loss solution for adults with obesity and a body mass index of 30 to 40 kg/m2.
The San Carlos, Calif.-based company said that the device is approved for up to 12 months of treatment, which includes lifestyle modification counseling intended to help patients develop and maintain healthier habits.
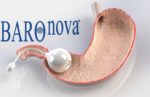
The newly cleared TransPyloric Shuttle device is designed to be delivered and retrieved endoscopically and uses a mechanical structure to maintain its shape and keep it in the stomach.
The constructed TPS device is approximately 5.6 cm in diameter, which BaroNova said is 85% to 90% smaller than fluid filled balloons. The system is intended to slow the passage of food and make a patient feel full sooner and stay full longer, the company said.
“FDA approval is a significant milestone for the company. The TPS device is unique in its treatment efficacy and duration. Further validation of potential comorbidity benefits, like cardiometabolic improvement observed in the pivotal study, during the post-market phase may translate to important advantages in achieving healthcare coverage for the TPS device in the future. We look forward to bringing this new technology to patients and physicians,” clinical & regulatory affairs senior VP Dr. Lian Cunningham said in a press release.
Approval came based on data from the 302-patient ENDObesity II trial of the device which analyzed the safety and effectiveness of the TransPyloric Shuttle Device in treating obesity in patients with a body mass index of between 30-40kg/m2.
The trial met its primary endpoints of the percent of total body weight loss at one year follow-up post-procedure and the proportion of people in the treatment group who achieved 5% total body weight loss or more at one year, BaroNova said.
“Those of us who participated in the pivotal trial have the firsthand experience with the TPS device and were pleased with its patient outcomes. In addition to the clinically meaningful weight loss seen in the study group who received the device, we also observed cardio-metabolic improvements in that cohort. We are excited to see that the device is approved by the FDA and look forward to using it in our clinical practice,” ENDObesity II study lead investigator Dr. Richard Rothstein of the Dartmouth Geisel School of Medicine said in a prepared release.
“The vast majority of patients with obesity are left untreated today. Endoscopically delivered intragastric devices can help close the obesity treatment gap and offer alternative options for qualified patients who are not eligible, or unwilling, to undergo metabolic and bariatric surgery. The TPS device design addresses some of the limitations with the first-generation intragastric devices and offers longer treatment duration which is clinically attractive,” ENDObesity II study principal investigator Dr. Wayne English of the Vanderbilt University Weight Loss Center said in a prepared statement.
Last November, Baronova released the primary results from the pivotal ENDObesityII study of its TransPyloric Shuttle device intended to treat obesity.

