Updated August 30, 2013, at 4:00 p.m. with comments from Stryker.

The FDA issued a Class I recall for 5 lots of Stryker Spine’s Oasys Midline Occipital Plate, warning patients and physicians that the device might fracture after surgery.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
Updated August 30, 2013, at 4:00 p.m. with comments from Stryker.

The FDA issued a Class I recall for 5 lots of Stryker Spine’s Oasys Midline Occipital Plate, warning patients and physicians that the device might fracture after surgery.

Tissue Regeneration Systems won FDA clearance for its 3D-printed Cranial Bone Void Filler, the flagship product in a suite of devices indicated for cranial reconstruction.
Designed to fill holes left behind after surgery, the CBV filler appears to be the 1st 3D-printed device of its kind to get the FDA’s green light for the U.S. market.


Cardiovascular device firm PFM Medical has the FDA’s green light to market its Nit-Occlud PDA permanent prosthesis in the U.S., according to an approval order posted on FDA’s Website this month.


San Francisco-based AliveCor Inc. last week launched the visibility-enhancing AliveECG App for its iPhone-based Heart Monitor ECG, an add-on that the company says will "improve the visual output" of the device.
The app uses an "
Enhanced Filter," a technology that reduces noise from the environment or muscle vibrations, which can cause ECG strips to be difficult to read, according to the privately held company.

Shares of HeartWare International (NSDQ:HTWR) are on watch in the wake of negative commentary from a Northland Capital Markets analyst.
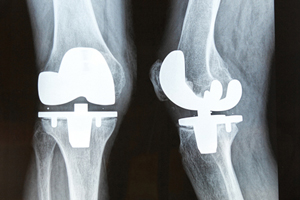
The societal and economic savings of knee replacement surgery outweigh its costs nearly 2-fold, according to an American Academy of Orthopaedic Surgeons study.

The FDA’s Science Board yesterday agreed to form a new subcommittee to help the agency examine 3 specified domains of regulatory science activities related to medical devices, assess progress made since 2007, and address the challenges posed by rapidly evolving science and technology.
These challenges include the scientific implications of the increasing globalization of product development and supply chains, the federal watchdog agency noted in background materials released ahead of the meeting.

Symbios Medical Products last week recalled all of its GoPump rapid recovery systems and GOBlock kits manufactured with flow control components assembled prior to July 2012, according to the FDA.
These products were found to potentially cause excessively high flow rates, the agency reported. Due to the rapid influx of medication, particularly in patients with low body mass or advanced age, patient toxicity and serious injury including seizure, dysrhythmia and death can result, the FDA noted.

A panel of experts today recommended that FDA lower the regulatory bar for pedicle screw spinal systems, which are intended to treat degenerative disc disease and a limited type of spondylolisthesis, or shifting of vertebra in response to a vertebral stress fracture.

