
An FDA panel today voted today to recommend approval for Staar Surgical’s (NSDQ:STAA) novel Visian Toric implantable collamer lens for treatment of myopic astigmatism in adults aged 21-45.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

An FDA panel today voted today to recommend approval for Staar Surgical’s (NSDQ:STAA) novel Visian Toric implantable collamer lens for treatment of myopic astigmatism in adults aged 21-45.

The FDA’s Orthopedic & Rehabilitation Devices advisory panel last week recommended that the watchdog agency classify spinal sphere devices into its highest risk Class III category because there is insufficient evidence of their safety and effectiveness when used in intervertebral body fusion procedures.
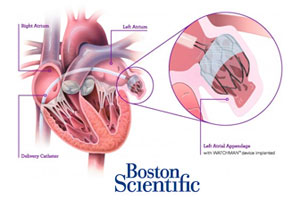
The FDA’s Circulatory Systems Devices Panel voted 13-1 yesterday to recommend approval of Boston Scientific‘s (NYSE:BSX) Watchman heart implant for reducing the risk of stroke and systemic embolism.

The FDA’s Circulatory Systems Devices Panel today voted 13-1 to recommend approval of Boston Scientific’s(NYSE:BSX) Watchman 1st-of-its-kind left atrial appendage (LAA) closure device for reducing the risk of stroke and systemic embolism.
Updated October 10, 2013, at 5 p.m. with comments from the CDRH panel.


An FDA advisory panel voted yesterday that Ocular Therapeutix’s ReSure topical hydrogel post-op sealant is safe, but split on questions of efficacy and whether the benefits of the treatment outweigh its risks.
The FDA’s Ophthalmic Devices Panel voted 9-1, with 1 abstention, that the ReSure sealant is safe; 5-3, with 3 abstentions, that it is effective; and 5-1, with 5 abstentions, that its benefits outweigh its risks.

A panel of experts today voted that Ocular Therapeutix’s ReSure Sealant topical hydrogel post-op sealant is safe, but panelists were split on questions of efficacy and whether the benefits of the treatment outweigh the risks.

Jan Medical is partnering with software-driven medtech firm Brainlab to gain FDA clearances via 2 clinical trials, the companies announced.
As part of the collaboration, Brainlab will add a representative to Jan Medical’s board, the companies said in a press release.
Mountain View, Calif.-based Jan Medical makes the the Nautilus NeuroWave, a portable, non-invasive brain sensing system designed to detect abnormal neurological conditions, including ischemic stroke, vasospasm, and concussion.

Heart failure patients with implant-based remote monitoring die less frequently from any cause, according to Biotronik study results released at the European Society of Cardiology congress in Amsterdam.

