
By Deborah Bell, executive director of business development in Australia, Avania
Whether to control costs or to recruit a larger population, medical device manufacturers are looking beyond the U.S., the EU, and Asia to conduct clinical trials. When planning global studies and early commercialization, Australia is a strongly considered market. The access to innovative medical services as well as an established healthcare infrastructure dedicated to excellence make the region an attractive, yet often unthought of, clinical research location.
While most manufacturers recognize the country’s market potential, its reimbursement process is rigorous and can seem multifactorial or complicated to the uninitiated. Here, we break down the opportunities available for both conducting clinical trials and achieving successful commercialization.
Why Australia?
Aside from beautiful beaches and good wine, Australia boasts a robust clinical research sector and a government committed to its growth. While the country’s public health insurance program — Medicare — does not offer reimbursement for medical device clinical trials, it’s well worth considering for early clinical research.
- One of the best healthcare systems in the world. The Commonwealth Fund ranked Australia’s public-private system third in the world, with top marks in health equity and outcomes. Many of its physicians have studied or trained in the U.S. and U.K., and many of its clinical practices resemble practices in those countries.
- Top-tier research facilities. Australia is internationally known for its skilled research teams and state-of-the-art medical processes, procedures, and equipment. The country adheres to good clinical practice (GCP) standards, which means its data are accepted by most regulatory agencies, including the FDA. International Conference on Harmonisation (ICH) and International Organization for Standardization (ISO) GCP standards are mandatory for all clinical trials conducted in Australia.
- A smooth regulatory pathway. Australia’s two-tier healthcare system allows for a fluid and fairly seamless process for proof-of-concept through clinical trials. The collaborative network of public institutional and independent ethics committees provides a streamlined, centralized review of clinical trials to accelerate approval times and consolidate requisite documentation.
- Significant tax incentives. The Australian government provides a research and development tax incentive to businesses investing in R&D activities, including clinical trials. Businesses with annual revenues of less than $20 million AUS receive a 45% tax offset, while businesses with higher revenue receive a 40% tax offset. Medical device manufacturers based outside of Australia should consider creating a business entity or subsidiary in Australia to take advantage of this benefit.
- A potentially faster process. As regulatory processes tend to resemble those in Europe, clinical trials tend to move faster than they would in the U.S. Study teams often block-book patients so physicians perform procedures on multiple patients a day, spread through multiple sites.
How to Get Started: Appoint a Sponsor
Companies considering conducting a clinical trial in Australia must have an Australian sponsor. That sponsor could be an individual physician, a healthcare facility, or a CRO. Sponsors work directly with the Therapeutic Goods Administration (TGA) to obtain approval to import unapproved product via a Clinical Trial Notification (CTN), to obtain regulatory approval, and to obtain commercial distribution. Local sponsors also work with clinical sites and ethics committees to gain ethical approval to conduct the study.
The Australian Government’s National Health and Medical Research Council (NHMRC) oversees medical device clinical trials. The organization provides research funding, distributes health evidence and clinical research guidance, and advises on health policy.
Reimbursement for Implantable Devices
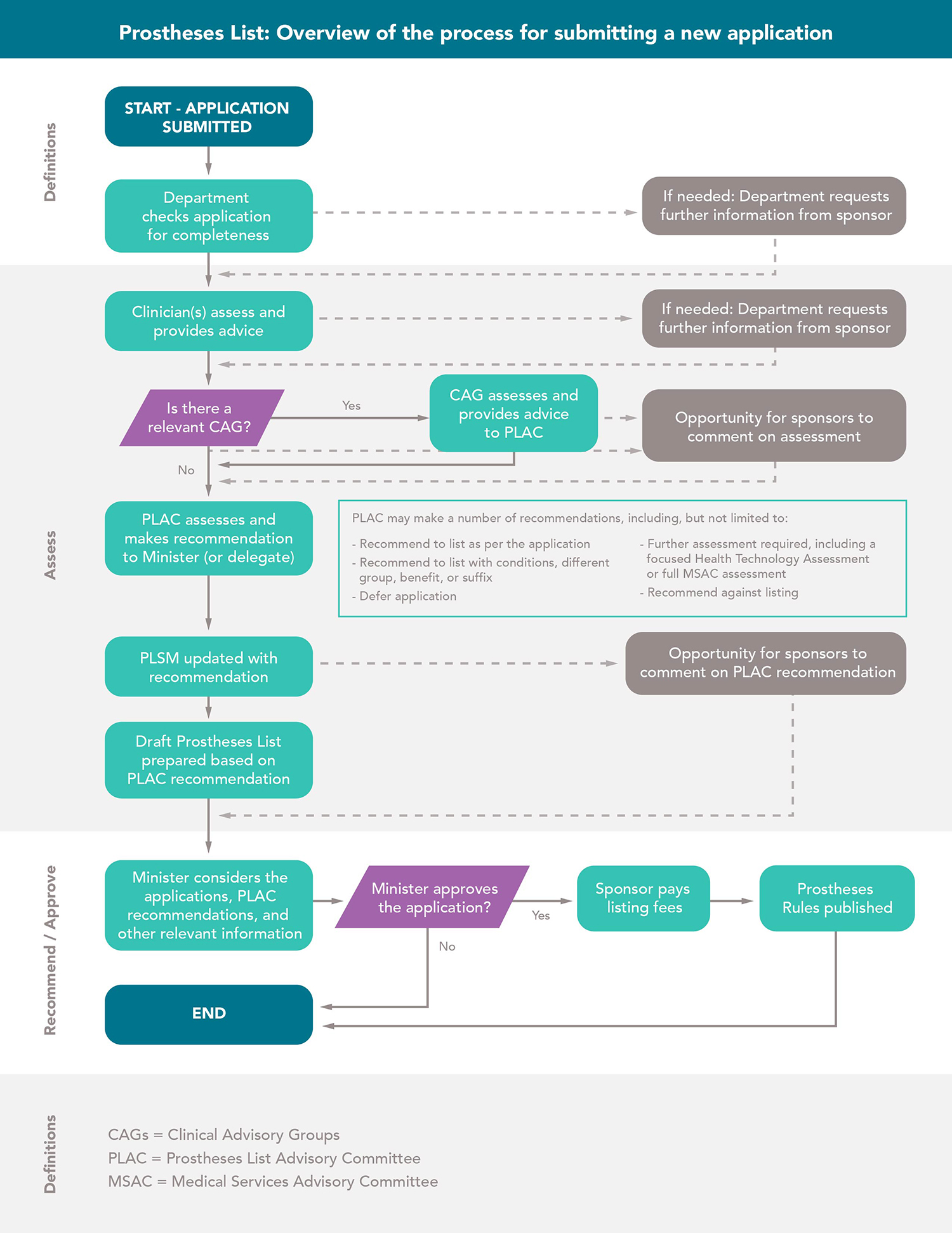
Companies bringing implantable devices into the Australian market can apply for inclusion on the prostheses list, which is governed by the Prostheses List Advisory Committee (PLAC). Private health insurers pay a specific benefit for devices on this list. The list currently includes more than 11,000 joint replacement devices, cardiac implantable devices, stents, infusion pumps, catheters, and cardiac remote monitoring systems.
Upon receipt of application, a Clinical Advisory Group (CAG) and a panel of clinical experts both assess products’ comparative clinical effectiveness based on the evidence submitted by sponsors. The CAGs and the expert panel advise the PLAC on whether a product satisfies the criteria for listing and suggest the grouping in which the product should be listed.
After the PLAC recommends a product for listing, the process moves to the government’s Minister of Health who decides whether to grant or deny an application. The entire process takes about six months.
How to Increase Your Odds of Reimbursement Approval
As with U.S. clinical trials, manufacturers must consider reimbursement early in the clinical trial planning process. When designing the protocol and determining what data to gather, factor in the following:
- Optimize clinical trial design. Design your clinical trial so it produces as much information as possible that’s as valuable as possible to regulatory and payer organizations. That includes quality, outcomes, and quality of life data.
- Focus less on price, more on value. Many U.S. manufacturers want to price their products at a premium because they are superior in ease of use and/or efficacy compared to similar products. That may be true, but Australia prioritizes value. Demonstrate how your product reduces healthcare costs and consider these metrics when planning clinical trials. The goal is to collect evidence in a single study that will help prove your case for reimbursement.
- Don’t plan to negotiate. Australia’s payer system offers a fixed price for medical devices. Unlike the U.S., it doesn’t negotiate. Hospital systems have a set budget to work with, which leaves little to no room for negotiation.
With its generous tax incentives, an excellent healthcare system, and a relatively smooth regulatory processes, it’s wise to keep Australia on the map when planning your next global trial.
Avania is an integrated global, full-service CRO with specialized expertise in medical device, novel technology, and combination products. They advance products from feasibility all the way through post-approval trials in analytics, clinical trials, consulting, regulatory, reimbursement, and more. To learn more, visit: avaniaclinical.com.
Sponsored content by Avania

