Medical device recalls for August 2014
September 3, 2014 by MassDevice

August Summary
Class I recalls: 244
Class II recalls: 171
Class I recalls
Date Posted, Recalling Manufacturer, Trade Name/Product, Reason for Recall Read more
![]()
![]()
Medtronic wins CE Mark for CoreValve Evolut R
September 3, 2014 by Brad Perriello
Medtronic said it won CE Mark approval for the latest version of its transcatheter aortic valve implant, the CoreValve Evolut R, meaning the repositionable replacement heart valve is now available on the European market.
The CoreValve Evolut R is designed to allow implanting physicians to return the device to its EnVeo R delivery catheter and reposition it as needed, according to a press release. Read more
![]()
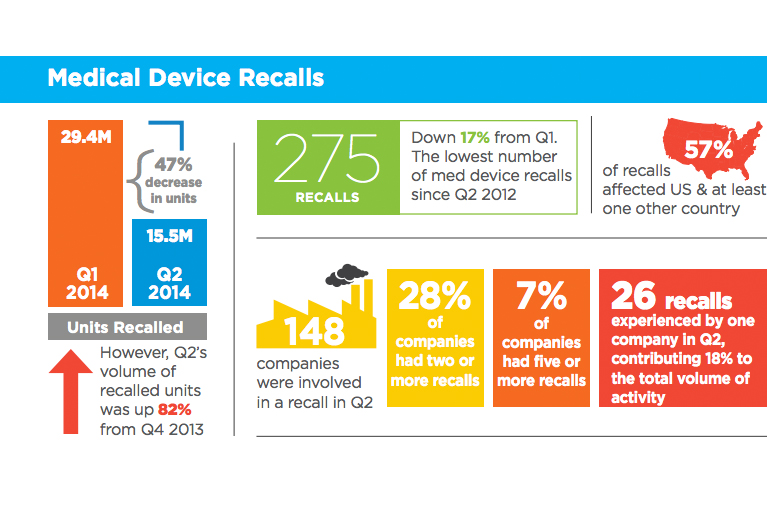
Medical device recalls hit 2-year low in Q2
September 5, 2014 by Brad Perriello
The number of medical device recalls fell 17% during the 2nd quarter to a 2-year low, with 47% fewer units pulled from shelves than during the same period last year, according to a report from Stericycle.
There were 275 recalls involving 148 companies during the quarter, the lowest number since the 2nd quarter of 2012, according to the report, which is compiled using data from the FDA and other regulatory agencies. The number of recalled units, however, rose 82% compared with the 4th quarter of 2013, the company said. Read more
![]()
FDA orders iRadimed to submit new 510(k) after site inspection, stock tanks
September 3, 2014 by Arezu Sarvestani

Florida-based iRadimed watched its stock nosedive today after revealing an FDA warning letter chiding the company for its complaint handling and demanding a new 510(k) application for its infusion pumps.
Agency inspectors ruled after a site inspection that iRadimed’s IV infusion pumps are on the market illegally because they include updated software that has not been reviewed by the proper authorities. The FDA added that iRadimed needs to seek separate 510(k) clearances for its mRidium 3860+ infusion pump, as it was deemed a significant departure from the mRidium 3860 and mRidium 3850 pumps. Read more
![]()
Diabetes: Medtronic wins Health Canada nod for enhanced Enlite glucose sensor
September 4, 2014 by Brad Perriello
Medtronic said today that Health Canada granted a license for the enhanced Enlite glucose sensor, part of the medical device company’s MiniMed Veo integrated insulin pump and continuous glucose monitoring system.
That means the enhanced Enlite sensor will be available to patients north of the border in early October, according to a press release. Read more