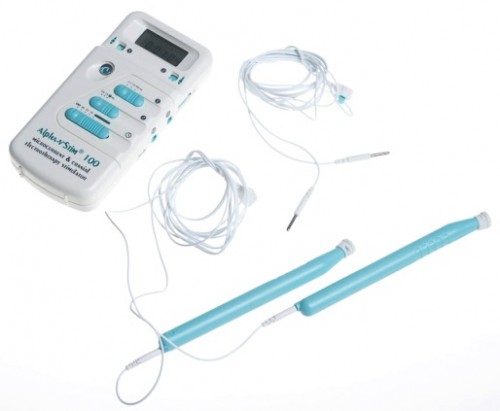
Alpha-Stim launched a campaign to change a proposed FDA ruling regarding pre-market approval for its cranial electrotherapy stimulation devices.
The FDA is proposing a rule that would require pre-market approval of CES devices, which have heretofore been cleared for the U.S. market via 510(k) process for the treatment of anxiety, depression, insomnia and pain.
Since the announcement, the federal watchdog agency has received more than 100 written objections by patients and medical professionals vouching for device safety, efficacy and lack of side effects on behalf of Alpha-Stim.
Sign up to get our free newsletters delivered straight to your inbox
According to the company, 99.9 percent of Alpha-Stim users consider the device effective.
Alpha-Stim’s CES technology applies mild electrical current to patients through electrodes that attach to the earlobes. The current coaxes the brain’s electrical signals to normalize and has been used by doctors, psychologists and in the military community.
Approved for over-the-counter sale in Europe, Canada and China, Alpha-Stim’s devices have gone through six extensive FDA review processes since 1981 and have been cleared to market.
The pre-market approval ruling would cost the EPI Inc. subsidiary $1 million for a product that the FDA approved more than 30 years ago, the company said.
EPI Inc. asked the federal watchdog agency to review all of Alpha-Stim’s safety data and revise the findings within the proposed rule.

