 Advanced Cooling Therapy said today that it won CE Mark approval in the European Union for use of its esophageal cooling device with Stryker‘s (NYSE:SYK) Altrix Precision Temperature Management System.
Advanced Cooling Therapy said today that it won CE Mark approval in the European Union for use of its esophageal cooling device with Stryker‘s (NYSE:SYK) Altrix Precision Temperature Management System.
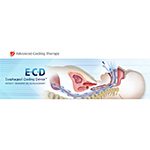
The Chicago-based company’s ECD is designed to modulate and control patient temperature using a single-use triple lumen system that is inserted into the patient’s esophagus. The device can be quickly inserted, like a standard gastric tube, and can control a patient’s temperature in a variety of clinical environments, including the operating room, emergency room and ICU.
Advanced Cooling Therapy said that it is the only product on the market approved to use the esophagus for temperature modulation.
“This new CE mark clearance addresses a significant need in a growing market, in which temperature management is increasingly important for both warming and cooling patients when clinically indicated,” international sales manager Markus Tödtling said in prepared remarks. “The ECD’s compatibility with a new line of control units, or heat exchangers, now available on the market, enables us to meet the growing needs of hospitals across Europe that are utilizing the Altrix Precision Temperature Management System in emergency departments, intensive care units, and operating rooms for a wide range of cooling and warming needs.”
“Because our existing product line is compatible with the majority of existing control units, the ECD is already being deployed in a growing number of hospitals worldwide,” North America VP of sales Robin Drassler added. The company is still waiting for FDA approval to use its ECD with Stryker’s Altrix system.
The ECD won FDA de novo clearance for its use with Stryker’s Medi-Therm III in June 2015. Last January, it won 510(k) clearance from the FDA for the ECD with Cincinnati Sub-Zero’s Blanketrol II and III Hyper-Hypothermia systems.
In May 2016, the company won expanded CE Mark approval in the E.U. for its ECD, clearing it for use for up to 120 hours.

