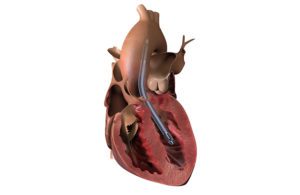
The study — published in the July edition of the American Society for Artificial Internal Organs (ASAIO) Journal — examined the outcomes of the first 55 patients treated with Impella 5.5 with SmartAssist at Cleveland Clinic, Hackensack University Medical Center/Hackensack Meridian Health and Cedars-Sinai Medical Center.
Study authors and cardiac surgeons Drs. Ed Soltesz, Mark Andersonv and Danny Ramzy cautioned that the study represented an early experience in a heterogeneous patient population, but they still found the results encouraging. They deemed the Impella 5.5 with SmartAssist to be safe and effective for a number of challenging clinical scenarios including AMI cardiogenic shock.
“This report demonstrates the benefit of unloading cardiogenic shock patients, and it is encouraging to see real-world data demonstrate improved survival rates with use of Impella 5.5 with SmartAssist,” said Ramzy, assistant professor and director of robotic and minimally invasive cardiac surgery at Cedars-Sinai Medical Center.
“The continued use of best practices, techniques and innovative technology like Impella allows us to provide better patient care that improves outcomes and quality of life,” Ramzy said in a news release.
FDA in September 2019 approved the Impella 5.5 with SmartAssist for treatment of cardiogenic shock for up to 14 days.

