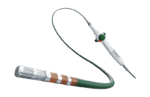
 Abbott (NYSE:ABT) today announced it has enrolled the first participants in its TactiFlex PAF IDE study to evaluate the performance of the TactiFlex sensor-enabled ablation catheter.
Abbott (NYSE:ABT) today announced it has enrolled the first participants in its TactiFlex PAF IDE study to evaluate the performance of the TactiFlex sensor-enabled ablation catheter.
The catheter is designed to treat people suffering from paroxysmal atrial fibrillation (PAF) with symptoms that are unable to be managed by medication.
TactiFlex combines Abbott’s contact force-sensing technology within the TactiCath SE catheter with a catheter tip design found in the company’s Flexibility irrigated ablation catheter.
Get the full story on our sister site, Medical Tubing + Extrusion.

